Modulating Effects of Fimasartan and Omega-3 on Cisplatin-Induced Testicular Toxicity in Rats
DOI:
https://doi.org/10.54133/ajms.v10i2.2691الكلمات المفتاحية:
Antioxidant، Cisplatin، Fimasartan، Omega-3الملخص
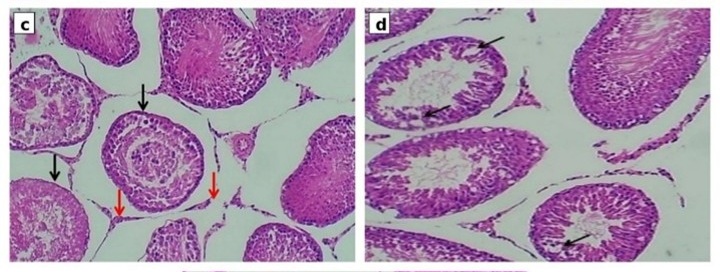
Background: Cisplatin is a widely used antineoplastic drug in different types of cancers (ovarian, testicular, and hematological) with several types of adverse effects, including testicular toxicity. Fimasartan is a newer angiotensin-receptor blocker (ARB) that has antioxidant and anti-inflammatory properties. Omega-3 is an unsaturated fatty acid that has antioxidant and anti-inflammatory effects. Objective: to evaluate the protective effects of fimasartan alone or in combination with omega-3 against cisplatin-induced testicular toxicity. Methods: Thirty Wistar rats were divided into five groups: control group, cisplatin-treated group, fimasartan+cisplatin group, fimasartan+omega-3+cisplatin group, and omega-3+cisplatin group. Treatments were administered for 10 consecutive days. On day 10, a single intraperitoneal dose of cisplatin (7mg/kg) was given to induce testicular toxicity. On day 11, animals were sacrificed. Testicular tissue homogenates were used to measure malondialdehyde (MDA), reduced glutathione (GSH), and superoxide dismutase (SOD). Serum levels of testosterone and inhibin-B were measured using ELISA. Histopathological examination of the testes was also performed. Results: Cisplatin administration significantly increased MDA levels and significantly decreased GSH, SOD, testosterone, and inhibin-B levels compared with the control group. Treatment with fimasartan alone or in combination with omega-3 significantly attenuated these alterations and improved histopathological changes in testicular tissue. Conclusions: Fimasartan exerts protective effects against cisplatin-induced testicular toxicity through its antioxidant and reducing oxidative stress effects, and its combination with omega-3 enhances these protective effects.
التنزيلات
المراجع
Li L, Lin W, Wang Z, Huang R, Xia H, Li Z, et al. Hormone regulation in testicular development and function. Int J Mol Sci. 2024;25(11):5805. doi: 10.3390/ijms2511580. DOI: https://doi.org/10.3390/ijms25115805
Antar SA, Halouani A, Gad C, Al-Karmalawy AA. An overview of the mechanisms of cadmium-induced toxicity in the male reproductive system. Pharm Sci. 2024;30:36–53. doi: 10.34172/PS.2023.10. DOI: https://doi.org/10.34172/PS.2023.10
Bhardwaj JK, Panchal H, Saraf P. Cadmium as a testicular toxicant: A review. J Appl Toxicol. 2021;41:105–117. doi: 10.1002/jat.4052. DOI: https://doi.org/10.1002/jat.4055
Almohammadi A, Alqarni A, Alraddadi R, Alzahrani F. Assessment of patients’ knowledge in managing side effects of chemotherapy: Case of King Abdul-Aziz University Hospital. J Cancer Educ. 2020;35(2):334–338. doi: 10.1007/s13187-019-01605-4. DOI: https://doi.org/10.1007/s13187-019-1469-2
Alafifi S, Wahdan S, Elsherbiny D, Azab S. Doxorubicin-induced testicular toxicity: Possible underlying mechanisms and promising pharmacological treatments in experimental models. Arch Pharm Sci Ain Shams Univ. 2022;6(2):196–207. doi: 10.21608/aps.2022.155127.1098. DOI: https://doi.org/10.21608/aps.2022.155127.1098
Howell SJ, Shalet SM. Testicular function following chemotherapy. Hum Reprod Update. 2001;7(4):363–369. doi: 10.1093/humupd/7.4.363. DOI: https://doi.org/10.1093/humupd/7.4.363
Tchounwou PB, Dasari S, Noubissi FK, Ray P, Kumar S. Advances in our understanding of the molecular mechanisms of action of cisplatin in cancer therapy. J Exp Pharmacol. 2021;13:303–328. doi: 10.2147/JEP.S267383. DOI: https://doi.org/10.2147/JEP.S267383
Abdel-Latif R, Fathy M, Anwar HA, Naseem M, Dandekar T, Othman EM. Cisplatin-induced reproductive toxicity and oxidative stress: Ameliorative effect of kinetin. Antioxidants. 2022;11(5):915. doi: 10.3390/antiox11050915. DOI: https://doi.org/10.3390/antiox11050863
Fuertes MA, Alonso C, Pérez JM. Biochemical modulation of cisplatin mechanisms of action: Enhancement of antitumor activity and circumvention of drug resistance. Chem Rev. 2003;103(3):645–662. doi: 10.1021/cr020010d. DOI: https://doi.org/10.1021/cr020010d
Gandin V, Hoeschele JD, Margiotta N. Cisplatin in cancer therapy: Molecular mechanisms of action 3.0. Int J Mol Sci. 2023;24. doi: 10.3390/ijms24097917. DOI: https://doi.org/10.3390/ijms24097917
Marullo R, Werner E, Degtyareva N, Moore B, Altavilla G, Ramalingam SS, et al. Cisplatin induces a mitochondrial-ROS response that contributes to cytotoxicity. PLoS One. 2013;8(11):e81162. doi: 10.1371/journal.pone.0081162. DOI: https://doi.org/10.1371/journal.pone.0081162
Lee HY, Oh BH. Fimasartan: A new angiotensin receptor blocker. Drugs. 2016;76(10):1015–1022. doi: 10.1007/s40265-016-0597-2. DOI: https://doi.org/10.1007/s40265-016-0592-1
Drugs C, Data A, Bv I. Fimasartan. Am J Cardiovasc Drugs. 2011;11(4):249–252. doi: 10.1007/s40265-016-0592-1. DOI: https://doi.org/10.2165/11533640-000000000-00000
Kim TW, Yoo BW, Lee JK, Kim JH, Lee KT, Chi YH, et al. Synthesis and antihypertensive activity of pyrimidin-4(3H)-one derivatives as losartan analogues for new angiotensin II receptor type 1 (AT1) antagonists. Bioorg Med Chem Lett. 2012;22(4):1649–1654. doi: 10.1016/j.bmcl.2011.12.116. DOI: https://doi.org/10.1016/j.bmcl.2011.12.116
Nephrology U, Ospedaliera A, Riuniti O, Diseases R. The role of renin-angiotensin-aldosterone system in the progression of chronic kidney disease. Kidney Int. 2005;68:57–65. doi: 10.1111/j.1523-1755.2005.09911.x. DOI: https://doi.org/10.1111/j.1523-1755.2005.09911.x
Afroze S, Janakiraman AK, Gunasekaran B, Djearamane S, Wong LS. Potentials of omega-3 fatty acids as therapeutic drugs and its obstacles in the pathway: A critical review. J Pharm Pharmacogn Res. 2024;12:120–145. doi: 10.56499/jppres23.1689_12.1.120. DOI: https://doi.org/10.56499/jppres23.1689_12.1.120
Gareri P. Omega-3 long-chain polyunsaturated fatty acids in the elderly: A review. OBM Geriatr. 2022;6(2):1–28. doi: 10.21926/obm.geriatr.2202180. DOI: https://doi.org/10.21926/obm.geriatr.2202198
Hathaway D, Pandav K, Patel M, Riva-Moscoso A, Singh BM, Patel A, et al. Omega-3 acids and COVID-19: A comprehensive review. Infect Chemother. 2020;52:478–495. doi: 10.3947/ic.2020.52.4.478. DOI: https://doi.org/10.3947/ic.2020.52.4.478
O’Connell TD, Mason RP, Budoff MJ, Navar AM, Shearer GC. Mechanistic insights into cardiovascular protection for omega-3 fatty acids and their bioactive lipid metabolites. Eur Heart J Suppl. 2020;22:J3–J20. doi: 10.1093/eurheartj/suaa115. DOI: https://doi.org/10.1093/eurheartj/suaa115
Ateşşahin A, Karahan I, Türk G, Gür S, Yilmaz S, Çeribaşi AO. Protective role of lycopene on cisplatin-induced changes in sperm characteristics, testicular damage and oxidative stress in rats. Reprod Toxicol. 2006;21(1):42–47. doi: 10.1016/j.reprotox.2005.06.001. DOI: https://doi.org/10.1016/j.reprotox.2005.05.003
Chang SA, Lim BK, Lee YJ, Hong MK, Choi JO, Jeon ES. A novel angiotensin type I receptor antagonist, fimasartan, prevents doxorubicin-induced cardiotoxicity in rats. J Korean Med Sci. 2015;30(5):559–568. doi: 10.3346/jkms.2015.30.5.559. DOI: https://doi.org/10.3346/jkms.2015.30.5.559
Awad MM, Abd-Ellatif RN, Ibrahim S, Abd Elmaaboud MA, El-Shaer RA. Role of heme oxygenase (HO)-1 enzyme in the protective and therapeutic effect of omega-3 fatty acids on cisplatin-induced hepatic and renal toxicity in rats. Suez Canal Univ Med J. 2023;26(1):17–39. doi: 10.21608/scumj.2023.283960. DOI: https://doi.org/10.21608/scumj.2023.283960
Wang L, He Y, Li Y, Pei C, Olatunji OJ, Tang J, et al. Protective effects of nucleosides-rich extract from Cordyceps cicadae against cisplatin-induced testicular damage. Chem Biodivers. 2020;17(11). doi: 10.1002/cbdv.202000462. DOI: https://doi.org/10.1002/cbdv.202000671
Park HJ, Kim JS, Lee R, Song H. Cisplatin induces apoptosis in mouse neonatal testes organ culture. Int J Mol Sci. 2022;23(21). doi: 10.3390/ijms232113408. DOI: https://doi.org/10.3390/ijms232113360
Pogach LM, Gould S, Fshuang P. Characterization of cis-platinum-induced Sertoli cell dysfunction in rodents. Toxicol Appl Pharmacol. 1989;98:350–365. doi: 10.1016/0041-008X(89)90239-1. DOI: https://doi.org/10.1016/0041-008X(89)90239-1
Stewart J, Turner KJ. Inhibin B as a potential biomarker of testicular toxicity. Cancer Biomark. 2005;1:75–91. doi: 10.3233/CBM-2005-1109. DOI: https://doi.org/10.3233/CBM-2005-1109
Soni KK, Zhang LT, You JH, Lee SW, Kim CY, Cui WS, et al. The effects of MOTILIPERM on cisplatin-induced testicular toxicity in Sprague-Dawley rats. Cancer Cell Int. 2015;15:121. doi: 10.1186/s12935-015-0269-6. DOI: https://doi.org/10.1186/s12935-015-0274-1
Ferreiro ME, Amarilla MS, Glienke L, Méndez CS, González C, Jacobo PV, et al. Inflammatory mediators TNFα and nitric oxide arrest spermatogonia GC-1 cell cycle. Reprod Biol. 2019;19(4):329–339. doi: 10.1016/j.repbio.2019.10.002. DOI: https://doi.org/10.1016/j.repbio.2019.11.001
Brozovic A, Ambriović-Ristov A, Osmak M. Relationship between cisplatin-induced ROS, glutathione and BCL-2 and resistance to cisplatin. Crit Rev Toxicol. 2010;40:347–359. doi: 10.3109/10408441003601836. DOI: https://doi.org/10.3109/10408441003601836
Kim S, Kim SJ, Yoon HE, Chung S, Choi BS, Park CW, et al. Fimasartan protects against renal inflammation and fibrosis in mice with unilateral ureteral obstruction: The possible role of Nrf2. Int J Med Sci. 2015;12(11):891–904. doi: 10.7150/ijms.13187. DOI: https://doi.org/10.7150/ijms.13187
Roberts AW. Therapeutic development and current uses of BCL-2 inhibition. Hematology Am Soc Hematol Educ Program. 2020;2020(1):1–9. doi: 10.1182/hematology.2020000154. DOI: https://doi.org/10.1182/hematology.2020000154
Abbas W, Altemimi M, Qassam H, Hameed AA, Zigam Q, Abbas L, et al. Fimasartan ameliorates renal ischemia reperfusion injury via modulation of oxidative stress, inflammatory and apoptotic cascades in a rat model. J Med Life. 2022;15(2):241–251. doi: 10.25122/jml-2021-0154. DOI: https://doi.org/10.25122/jml-2021-0154
Li W, Khor TO, Xu C, Shen G, Jeong WS, Yu S, et al. Activation of Nrf2-antioxidant signaling attenuates NF-κB inflammatory response and elicits apoptosis. Biochem Pharmacol. 2008;76(11):1485–1499. doi: 10.1016/j.bcp.2008.07.029. DOI: https://doi.org/10.1016/j.bcp.2008.07.017
Cho JH, Choi SY, Ryu HM, Oh EJ, Yook JM, Ahn JS, et al. Fimasartan attenuates renal ischemia-reperfusion injury by modulating inflammation-related apoptosis. Korean J Physiol Pharmacol. 2018;22(6):661–670. doi: 10.4196/kjpp.2018.22.6.661. DOI: https://doi.org/10.4196/kjpp.2018.22.6.661
Chung S, Yoon HE, Kim SJ, Koh ES, Hong YA, et al. Oleanolic acid attenuates renal fibrosis in mice with unilateral ureteral obstruction via facilitating nuclear translocation of Nrf2. Nutr Metab. 2014;11:2. doi: 10.1186/1743-7075-11-2 DOI: https://doi.org/10.1186/1743-7075-11-2
Ryu S, Shin JS, Cho YW, Kim HK, Paik SH, Lee JH, et al. Fimasartan, an anti-hypertension drug, suppressed inducible nitric oxide synthase expression via NF-κB and AP-1 inactivation. Biol Pharm Bull. 2013;36:467–474. doi: 10.1248/bpb.b12-00859. DOI: https://doi.org/10.1248/bpb.b12-00859
Alramadneh T, Abu-Harirah HA, Saleh IA, Athamneh RY, Abusalah M, Okla MK, et al. Protective effects of omega-3 fatty acids against cisplatin-induced hepatocellular damage and oxidative stress in rats. Res Square. 2023. doi: 10.21203/rs.3.rs-3645657/v1. DOI: https://doi.org/10.21203/rs.3.rs-3645657/v1
Al-Attar AM, Al-Rethea HA. Chemoprotective effect of omega-3 fatty acids on thioacetamide-induced hepatic fibrosis in male rats. Saudi J Biol Sci. 2017;24(4):956–965. doi: 10.1016/j.sjbs.2016.01.029. DOI: https://doi.org/10.1016/j.sjbs.2016.01.029
Adeyemi WJ, Olayaki LA. Diclofenac-induced hepatotoxicity: Low dose of omega-3 fatty acids have more protective effects. Toxicol Rep. 2018;5:90–95. doi: 10.1016/j.toxrep.2017.12.002. DOI: https://doi.org/10.1016/j.toxrep.2017.12.002
Eraky SM, Abo El-Magd NF. Omega-3 fatty acids protect against acetaminophen-induced hepatic and renal toxicity in rats through HO-1–Nrf2–BACH1 pathway. Arch Biochem Biophys. 2020;686:108387. doi: 10.1016/j.abb.2020.10838740. DOI: https://doi.org/10.1016/j.abb.2020.108387
Kelsey TW, Miles A, Mitchell RT, Anderson RA, Wallace WH. A normative model of serum inhibin B in young males. PloS One. 2016;11(4):e0153843. doi: 10.1371/journal.pone.0153843. DOI: https://doi.org/10.1371/journal.pone.0153843
Guzelce EC, Galbiati F, Goldman AL, Gattu AK, Basaria S, Bhasin S. Accurate measurement of total and free testosterone levels for the diagnosis of androgen disorders. Best Pract Res Clin Endocrinol Metab. 2022;36(4):101683. doi: 10.1016/j.beem.2022.101683. DOI: https://doi.org/10.1016/j.beem.2022.101683
Didžiapetrienė J, Kazbarienė B, Tikuišis R, Dulskas A, Dabkevičienė D, Lukosevičienė V, et al. Oxidant/antioxidant status of breast cancer patients in pre-and post-operative periods. Medicina. 2020;56(2):70. doi: 10.3390/medicina56020070. DOI: https://doi.org/10.3390/medicina56020070
Cheng SB, Liu HT, Chen SY, Lin PT, Lai CY, Huang YC. Changes of oxidative stress, glutathione, and its dependent antioxidant enzyme activities in patients with hepatocellular carcinoma before and after tumor resection. PloS One. 2017;12(1):e0170016. doi: 10.1371/journal.pone.0170016. DOI: https://doi.org/10.1371/journal.pone.0170016
التنزيلات
منشور
كيفية الاقتباس
إصدار
القسم
الرخصة
الحقوق الفكرية (c) 2026 Al-Rafidain Journal of Medical Sciences

هذا العمل مرخص بموجب Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Published by Al-Rafidain University College. This is an open access journal issued under the CC BY-NC-SA 4.0 license (https://creativecommons.org/licenses/by-nc-sa/4.0/).










