Adapting Hydralazine as a Reliable Low-Toxicity Alternative to Cobalt Chloride for Hypoxia Modeling in Breast Cancer Cells
DOI:
https://doi.org/10.54133/ajms.v10i2.2860Keywords:
Breast cancer, Cobalt chloride, Hydralazine, Hypoxia, HIF-1α , MCF-7 cellsAbstract
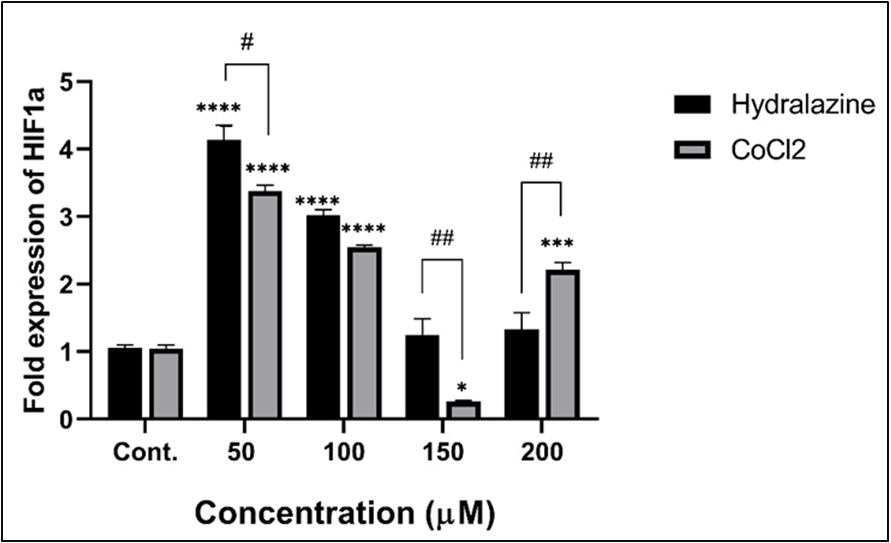
Background: Hypoxia represents a critical characteristic of the breast cancer microenvironment, responsible for tumor progression and therapeutic resistance. Although cobalt chloride (CoCl₂), which is used to induce cellular hypoxia, its application is restricted by severe cytotoxicity and off-target side effects. Objective: To evaluate hydralazine as an alternative for hypoxia induction in human MCF-7 breast cancer cells. Methods: MCF-7 cells were treated with increasing concentrations of hydralazine and CoCl₂ to compare the efficacy and cytotoxicity. Cell viability and stabilization of hypoxia-inducing factor-1 alpha (HIF-1α) were assessed using MTT and Western blotting assays, respectively. GAPDH protein was used as a loading control. Results: Hydralazine maintained near 98% cell viability across all tested doses up to 1600 µM at 24 hours, whereas CoCl₂ treatment resulted in dose-dependent cytotoxicity with a significant viability reduction to approximately 50% at the same maximum concentration. Furthermore, prolonged exposure (72 hours) to 1600 µM CoCl₂ caused severe cytotoxicity (<20% viability), while hydralazine-treated cells maintained significantly higher viability, enabling safer conditions for chronic hypoxia during long-term studies. Interestingly, Western blot results showed that hydralazine induced and stabilized HIF-1α protein, with a higher expression level than observed in CoCl₂-treated cells across the 50 to 150 µM concentration range. Conclusions: Hydralazine efficiently induces hypoxia in MCF-7 cells with favorable biocompatibility compared to CoCl₂ and can be a simple, safer, and more reliable chemical model for investigating signals mediated by HIF-1α in luminal breast cancer research, avoiding the variability of confounding heavy metal toxicity.
Downloads
References
Singh H. Role of molecular targeted therapeutic drugs in treatment of breast cancer: A review article. Glob Med Genet. 2023;10(02):079–86. doi: 10.1055/s-0043-57247. DOI: https://doi.org/10.1055/s-0043-57247
Qader MAJ, Numan IT, Al-Sudani BT, Abdalqader MA. Evaluation of 2-(4-Chlorophenyl)-4-(4-fluorophenyl)-5-pyridin-4-yl-1,2-dihydropyrazol-3-one as a p38 MAPK inhibitor in MCF-7 and MDA-MB-231 breast cancer cell lines. Al Mustansiriyah J Pharm Sci. 2025;25(3):317–326. doi: 10.32947/ajps.v25i3.1224. DOI: https://doi.org/10.32947/ajps.v25i3.1224
Galon J, Bruni D. Tumor immunology and tumor evolution: Intertwined histories. Immunity. 2020;52(1):55–81. doi: 10.1016/j.immuni.2019.12.018. DOI: https://doi.org/10.1016/j.immuni.2019.12.018
Obeagu EI. Oxygen deprivation in breast cancer: mechanisms, pathways, and implications. Ann Med Surg. 2025;87(6):3635–3659. doi: 10.1097/MS9.0000000000003334. DOI: https://doi.org/10.1097/MS9.0000000000003334
Xu R, Wang F, Yang H, Wang Z. Action sites and clinical application of HIF-1α inhibitors. Molecules. 2022;27(11):3426. doi: 10.3390/molecules27113426. DOI: https://doi.org/10.3390/molecules27113426
Qannita RA, Alalami AI, Harb AA, Aleidi SM, Taneera J, Abu-Gharbieh E, et al. Targeting hypoxia-inducible factor-1 (HIF-1) in cancer: Emerging therapeutic strategies and pathway regulation. Pharmaceuticals. 2024;17(2):195. doi: 10.3390/ph17020195. DOI: https://doi.org/10.3390/ph17020195
Abou Khouzam R, Zaarour RF, Brodaczewska K, Azakir B, Venkatesh GH, Thiery J, et al. The effect of hypoxia and hypoxia-associated pathways in the regulation of antitumor response: Friends or foes? Front Immunol. 2022;13:828875. doi: 10.3389/fimmu.2022.828875. DOI: https://doi.org/10.3389/fimmu.2022.828875
Capatina AL, Malcolm JR, Stenning J, Moore RL, Bridge KS, Brackenbury WJ, et al. Hypoxia-induced epigenetic regulation of breast cancer progression and the tumour microenvironment. Front Cell Dev Biol. 2024;12:1421629. doi: 10.3389/fcell.2024.1421629. DOI: https://doi.org/10.3389/fcell.2024.1421629
Ramachandramoorthy H, Dang T, Srinivasa A, Nguyen KT, Nguyen P. Development of a smart portable hypoxic chamber with accurate sensing, Control and visualization of in vitro cell culture for replication of cancer microenvironment. Cancers (Basel). 2023;15(14):3645. doi: 10.3390/cancers15143645. DOI: https://doi.org/10.3390/cancers15143645
Chatard M, Puech C, Perek N, Roche F. Hydralazine is a suitable mimetic agent of hypoxia to study the impact of hypoxic stress on in vitro blood-brain barrier model. Cell Physiol Biochem. 2017;42(4):1592–1602. doi: 10.1159/000479399. DOI: https://doi.org/10.1159/000479399
Tripathi VK, Subramaniyan SA, Hwang I. Molecular and cellular response of co-cultured cells toward cobalt chloride (CoCl2)-induced hypoxia. ACS Omega. 2019;4(25):20882–20893. doi: 10.1021/acsomega.9b01474. DOI: https://doi.org/10.1021/acsomega.9b01474
Li Q, Ma R, Zhang M. CoCl2 increases the expression of hypoxic markers HIF 1α, VEGF and CXCR4 in breast cancer MCF 7 cells. Oncol Lett. 2018;15(1):1119–1124. doi: 10.3892/ol.2017.7369. DOI: https://doi.org/10.3892/ol.2017.7369
Rana NK, Singh P, Koch B. CoCl2 simulated hypoxia induce cell proliferation and alter the expression pattern of hypoxia associated genes involved in angiogenesis and apoptosis. Biol Res. 2019;52(1):12. doi: 10.1186/s40659-019-0221-z. DOI: https://doi.org/10.1186/s40659-019-0221-z
Canhasi L, Tina E, Eremo AG. Hypoxia-mimetic by CoCl2 increases SLC7A5 expression in breast cancer cells in vitro. BMC Res Notes. 2023;16(1):366. doi: 10.1186/s13104-023-06650-2. DOI: https://doi.org/10.1186/s13104-023-06650-2
Knowles HJ, Tian YM, Mole DR, Harris AL. Novel Mechanism of Action for Hydralazine: induction of hypoxia-inducible factor-1alpha, vascular endothelial growth factor, and angiogenesis by inhibition of prolyl hydroxylases. Circ Res. 2004;95(2):162–169. doi: 10.1161/01.RES.0000134924.89412.70. DOI: https://doi.org/10.1161/01.RES.0000134924.89412.70
Romão IC, Siqueira SMC, Silva Abreu FOM da, dos Santos HS. Hydralazine and hydrazine derivatives: Properties, applications, and repositioning potential. Chem Biodivers. 2025;22(3):e202401561. doi: 10.1002/cbdv.202401561. DOI: https://doi.org/10.1002/cbdv.202401561
Ghasemi M, Turnbull T, Sebastian S, Kempson I. The MTT assay: Utility, limitations, pitfalls, and interpretation in bulk and single-cell analysis. Int J Mol Sci. 2021;22(23):12827. doi: 10.3390/ijms222312827. DOI: https://doi.org/10.3390/ijms222312827
Naser RA, Arif IS, Al-Sudani BT. Erastin induces ferroptosis and apoptosis in MDA-MB-231 breast cancer cell line. Al-Rafidain J Med Sci. 2025;8(2):168–172. doi: 10.54133/ajms.v8i2.1950. DOI: https://doi.org/10.54133/ajms.v8i2.1950
Sule R, Rivera G, Gomes AV. Western blotting (immunoblotting): History, theory, uses, protocol and problems. Biotechniques. 2023;75(3):99–114. doi: 10.2144/btn-2022-0034. DOI: https://doi.org/10.2144/btn-2022-0034
Ryu K, Park C, Lee Y. Hypoxia-inducible factor 1 alpha represses the transcription of the estrogen receptor alpha gene in human breast cancer cells. Biochem Biophys Res Commun. 2011;407(4):831–836. doi: 10.1016/j.bbrc.2011.03.119. DOI: https://doi.org/10.1016/j.bbrc.2011.03.119
Yang J, AlTahan A, Jones DT, Buffa FM, Bridges E, Interiano RB, et al. Estrogen receptor-α directly regulates the hypoxia-inducible factor 1 pathway associated with antiestrogen response in breast cancer. Proc Natl Acad Sci. 2015;112(49):15172–15177. doi: 10.1073/pnas.1422015112. DOI: https://doi.org/10.1073/pnas.1422015112
Jehanno C, Le Goff P, Habauzit D, Le Page Y, Lecomte S, Lecluze E, et al. Hypoxia and ERα transcriptional crosstalk is associated with endocrine resistance in breast cancer. Cancers (Basel). 2022;14(19):4934. doi: 10.3390/cancers14194934. DOI: https://doi.org/10.3390/cancers14194934
Lafi Z, Alshaer W, Gharaibeh L, Alqudah DA, AlQuaissi B, Bashaireh B, et al. Synergistic combination of doxorubicin with hydralazine, and disulfiram against MCF-7 breast cancer cell line. PLoS One. 2023;18(9):e0291981. doi: 10.1371/journal.pone.0291981. DOI: https://doi.org/10.1371/journal.pone.0291981
Yahyapour A, Askari N, Yaghoobi MM. Combination of hydralazine and all-trans retinoic acid targeting breast cancer cells. BMC Cancer. 2025;25(1):1427. doi: 10.1186/s12885-025-14477-2. DOI: https://doi.org/10.1186/s12885-025-14477-2
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Al-Rafidain Journal of Medical Sciences ( ISSN 2789-3219 )

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Published by Al-Rafidain University College. This is an open access journal issued under the CC BY-NC-SA 4.0 license (https://creativecommons.org/licenses/by-nc-sa/4.0/).











