The Correlation Between Thyroid Status, Family History, and Melasma Severity: A Cross-Sectional Dermatological Analysis
DOI:
https://doi.org/10.54133/ajms.v10i2.2820Keywords:
Anti-TPO, Family history, Melasma, mMASI, Thyroid autoimmunityAbstract
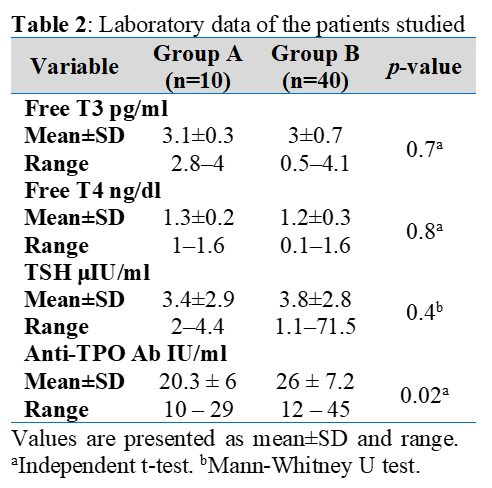
Background: Although family history is well recognized as a major predisposing factor in melasma, the extent to which hereditary background correlates with disease severity or underlying thyroid function abnormalities remains unclear. Objective: To assess the influence of family history on the severity of melasma and to determine if thyroid hormone levels or thyroid autoimmunity vary between women with and without a familial history of melasma. Methods: This cross-sectional study included 50 women diagnosed with facial melasma, who were stratified into two groups: those with a positive family history (n=10) and those without a family history (n=40). All participants underwent clinical evaluation using the modified Melasma Area and Severity Index (mMASI). Laboratory assessment included serum FT3, FT4, TSH, and anti-thyroid peroxidase (anti-TPO) antibodies measured via standardized immunoassays. Results: The mean mMASI scores were slightly lower in patients with a family history compared to those without, although the difference did not reach statistical significance (12.9±1.5 vs. 13.5±0.8; p=0.10). Thyroid hormone levels (FT3, FT4, TSH) showed no significant differences between groups. However, anti-TPO antibody levels were significantly higher in the non-familial group (26.0±7.2 vs. 20.3±6.0 IU/mL; p=0.02), suggesting increased thyroid autoimmunity in patients without hereditary predisposition. Conclusions: Family history did not significantly influence melasma severity or thyroid hormone levels. However, elevated anti-TPO titers among women without a family history indicate a potential autoimmune component in non-hereditary melasma.
Downloads
References
Wu IB, Lambert C, Lotti TM, Hercogová J, Sintim-Damoa A, Schwartz RA. Melasma. G Ital Dermatol Venereol. 2012;147(4):413-418. PMID: 23007216.
Nassar A, Abdelhamid AM, Samir MA. (2024). Melasma: Treatment modalities and new therapeutic strategies. J Comput Anal Appl. 2024;33(07):2403–2413.
Abdalla MA. Melasma clinical features, diagnosis, epidemiology and etiology: an update review. Siriraj Med J. 2021;73:841–850. doi: 10.33192/Smj.2021.109. DOI: https://doi.org/10.33192/Smj.2021.109
Espósito ACC, Cassiano DP, da Silva CN, Lima PB, Dias JAF, Hassun K, et al. Update on melasma—Part I: pathogenesis. Dermatol Ther (Heidelb). 2022;12:1967–1988. doi: 10.1007/s13555-022-00779-x. DOI: https://doi.org/10.1007/s13555-022-00779-x
Zhang J, Wang T, Li Z, Qin C, Dai J, Zhao Y, et al. Hormonal crosstalk in melasma: unraveling the dual roles of estrogen and progesterone in melanogenesis. Int J Mol Sci. 2025;26:10856. doi: 10.3390/ijms262210856. DOI: https://doi.org/10.3390/ijms262210856
Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201–1212. doi: 10.1111/ijd.17718. DOI: https://doi.org/10.1111/ijd.17718
Liu W, Chen Q, Xia Y. New mechanistic insights of melasma. Clin Cosmet Investig Dermatol. 2023;16:429–442. doi: 10.2147/CCID.S396272. DOI: https://doi.org/10.2147/CCID.S396272
Biró E, Szekanecz Z, Czirják L, Dankó K, Kiss E, Szabó NA, et al. Association of systemic and thyroid autoimmune diseases. Clin Rheumatol. 2006;25:240–245. doi: 10.1007/s10067-005-1165-y. DOI: https://doi.org/10.1007/s10067-005-1165-y
Pandya AG, Hynan LS, Bhore R, Riley FC, Guevara IL, Grimes P, et al. Reliability assessment and validation of the Melasma Area and Severity Index (MASI) and a new modified MASI scoring method. J Am Acad Dermatol. 2011;64:78–83. doi: 10.1016/j.jaad.2009.10.051. DOI: https://doi.org/10.1016/j.jaad.2009.10.051
Desai SR, Alexis AF, Elbuluk N, Grimes PE, Weiss J, Hamzavi IH, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269–279. doi: 10.1016/j.jaad.2023.07.1045. DOI: https://doi.org/10.1016/j.jaad.2023.07.1045
Handel AC, Miot LDB, Miot HA. Melasma: a clinical and epidemiological review. An Bras Dermatol. 2014;89:771–782. doi: 10.1590/abd1806-4841.20143063. DOI: https://doi.org/10.1590/abd1806-4841.20143063
Lutfi RJ, Fridmanis M, Misiunas AL, Pafume O, Gonzalez EA, Villemur JA, et al. Association of melasma with thyroid autoimmunity and other thyroidal abnormalities and their relationship to the origin of melasma. J Clin Endocrinol Metab. 1985;61:28–31. doi: 10.1210/jcem-61-1-28. DOI: https://doi.org/10.1210/jcem-61-1-28
Grimes P, Kelly AP, Torok H, Willis I. Community-based trial of a triple-combination agent for the treatment of facial melasma. Cutis. 2006;77:177–184. PMID: 16610738.
Kim EH, Kim YC, Lee ES, Kang HY. The vascular characteristics of melasma. J Dermatol Sci. 2007;46:111–116. doi: 10.1016/j.jdermsci.2007.01.009. DOI: https://doi.org/10.1016/j.jdermsci.2007.01.009
Thawabteh AM, Jibreen A, Karaman D, Thawabteh A, Karaman R. Skin pigmentation types, causes and treatment: a review. Molecules. 2023;28:4839. doi: 10.3390/molecules28124839. DOI: https://doi.org/10.3390/molecules28124839
Ogbechie-Godec OA, Elbuluk N. Melasma: an up-to-date comprehensive review. Dermatol Ther (Heidelb). 2017;7:305–318. doi: 10.1007/s13555-017-0194-1. DOI: https://doi.org/10.1007/s13555-017-0194-1
Miot LDB, Miot HA, Silva MG da, Marques MEA. Fisiopatologia do melasma. An Bras Dermatol. 2009;84:623–635. doi: 10.1590/S0365-05962009000600008. DOI: https://doi.org/10.1590/S0365-05962009000600008
Cohen B, Cadesky A, Jaggi S. Dermatologic manifestations of thyroid disease: a literature review. Front Endocrinol (Lausanne). 2023;14:1167890. doi: 10.3389/fendo.2023.1167890. DOI: https://doi.org/10.3389/fendo.2023.1167890
Razvi F, Ahmed FK. Melasma and thyroid autoimmunity: a case-control study to evaluate association, if any. Res J Med Sci. 2024;18:473–478. doi: 10.36478/makrjms.2024.11.473.478. DOI: https://doi.org/10.36478/makrjms.2024.11.473.478
Tekou A, Labbene I. Association of melasma with thyroid disorders. Sch J Appl Med Sci. 2023;11:1736–1742. doi: 10.36347/sjams.2023.v11i09.024. DOI: https://doi.org/10.36347/sjams.2023.v11i09.024
Baldini E, Odorisio T, Sorrenti S, Catania A, Tartaglia F, Carbotta G, et al. Vitiligo and autoimmune thyroid disorders. Front Endocrinol (Lausanne). 2017;8:290. doi: 10.3389/fendo.2017.00290. DOI: https://doi.org/10.3389/fendo.2017.00290
Duntas LH. Environmental factors and autoimmune thyroiditis. Nat Clin Pract Endocrinol Metab. 2008;4:454–460. doi: 10.1038/ncpendmet0896. DOI: https://doi.org/10.1038/ncpendmet0896
Ortonne JP, Arellano I, Berneburg M, Cestari T, Chan H, Grimes P, et al. A global survey of the role of ultraviolet radiation and hormonal influences in the development of melasma. J Eur Acad Dermatol Venereol. 2009;23:1254–1262. doi: 10.1111/j.1468-3083.2009.03295.x. DOI: https://doi.org/10.1111/j.1468-3083.2009.03295.x
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Al-Rafidain Journal of Medical Sciences ( ISSN 2789-3219 )

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Published by Al-Rafidain University College. This is an open access journal issued under the CC BY-NC-SA 4.0 license (https://creativecommons.org/licenses/by-nc-sa/4.0/).











