The Role of Fibroblast Growth Factor-2 and Caspase-3 in Nontoxic Multinodular Goiter versus Normal Thyroid Tissue Cases from Baghdad, Iraq
DOI:
https://doi.org/10.54133/ajms.v10i1.2778Keywords:
Active caspase-3, Fibroblast Growth Factor-2, Immunohistochemistry, Nontoxic multinodular goiter, Thyroid glandAbstract
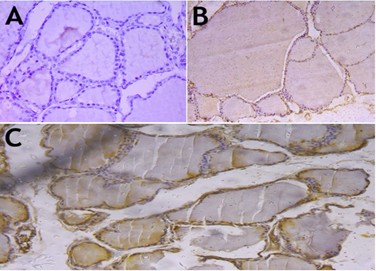
Background: Nontoxic multinodular (MNG) disease is one of the most prevalent thyroid pathologies, especially in endemic areas like Baghdad, Iraq. Objective: To determine the prognostic values of FGF-2 and active CASP-3 expression in cases of MNG to predict the possibility of cancer development. Methods: Fifty patients were included in the study in a retrospective design and were obtained from Al-Yarmouk Teaching Hospital in Baghdad, Iraq. Paraffin blocks were obtained, and histological procedures for slide preparation for light microscopy were employed. Immunohistochemical procedures to detect the expression of FGF2 and active CASP-3 markers were applied. Statistical analysis to predict the extent, intensity, total score, and H-score of the immunohistochemical results was done. Results: Analysis of cases showed an overexpression of the FGF2 marker in cases of MNG, and that was statistically significant. On the other hand, CASP-3 was downregulated in MNG cases, with an intensity of 0 in most instances, while a few cases showed intensities of 1+ and 2+, which were statistically significant. The H-score, on the other hand, was highly raised in MNG expressing FGF2; in contrast to CASP-3, which appeared very low in MNG tissues, and both results were of statistical significance. Conclusions: Overexpression of FGF2 and low expression of active CASP-3 in cases of MNG confirm the benign behavior of the disease, but it does not exclude the possibility of cancer development. This expression can be regarded as prognostic for early detection of tumor transition in benign tissue.
Downloads
References
Maniakas A, Zafereo M, Cabanillas ME. Anaplastic thyroid cancer: New horizons and challenges. Endocrinol Metab Clin North Am. 2022 ;51(2):391-401. doi: 10.1016/j.ecl.2021.11.020. DOI: https://doi.org/10.1016/j.ecl.2021.11.020
Abdulateef HH, Shukri EG, Ahmed BS. Immunohistochemical co-expression of nanog and oct4 in normal tissue around papillary thyroid cancer and multinodular goiter. Biochem Cell Arch. 2020; 20(2):4477–4485. Available at: www.connectjournals.com/bca
Dauksiene D, Petkeviciene J, Klumbiene J, Verkauskiene R, Vainikonyte-Kristapone J, Seibokaite A, et al. Factors associated with the prevalence of thyroid nodules and goiter in middle-aged euthyroid subjects. Int J Endocrinol. 2017;2017:8401518. doi: 10.1155/2017/8401518. DOI: https://doi.org/10.1155/2017/8401518
Can AS, Nagalli S. Substernal Goiter. 2025 Jan 15. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 32491348.
How J, Tabah R. Explaining the increasing incidence of differentiated thyroid cancer. CMAJ. 2007;177(11):1383-1384. doi: 10.1503/cmaj.071464. DOI: https://doi.org/10.1503/cmaj.071464
Rashid S, Alhiti A. Goiters in some Iraqi females. Alq J Med App Sci. 2022;5(2),479–482. doi: 10.5281/zenodo.7114227.
Atiea HA, Majeed NS, Abid-Saaidan M. The outcome of surgical treatment of fifty patients with goiter -n Baghdad-Iraq. Int J Adv Res. 2020;8(1):523-527. doi: 10.21474/ijar01/10324. DOI: https://doi.org/10.21474/IJAR01/10324
AlSaedi AH, Almalki DS, ElKady RM. Approach to thyroid nodules: Diagnosis and treatment. Cureus. 2024;16(1):e52232. doi: 10.7759/cureus.52232. DOI: https://doi.org/10.7759/cureus.52232
Abdulateef HH, Shukri EG, Ahmed BS. Immunohistochemical expression of ALHD1A1 in thyroid goiter and normal peritumor tissue around papillary thyroid carcinoma in a sample of Iraq patients. Ann Tropical Med Public Health. 2020;23(13):231347. doi: 10.36295/ASRO.2020.231347. DOI: https://doi.org/10.36295/ASRO.2020.231347
Kutanzi KR, Lumen A, Koturbash I, Miousse IR. Pediatric exposures to ionizing radiation: Carcinogenic considerations. Int J Environ Res Public Health. 2016;13(11):1057. doi: 10.3390/ijerph13111057. DOI: https://doi.org/10.3390/ijerph13111057
Unlu MT, Kostek M, Aygun N, Isgor A, Uludag M. Non-toxic multinodular goiter: From etiopathogenesis to treatment. Sisli Etfal Hastan Tip Bul. 2022;56(1):21-40. doi: 10.14744/SEMB.2022.56514. DOI: https://doi.org/10.14744/SEMB.2022.56514
CN A, Devani R, Jayaprakash H. Histomorphological patterns of thyroid lesions: A study from a tertiary care teaching hospital of Dr BR Ambedkar Medical College. Indian J Pathol Oncol. 2017;4(4):529-532.
Campbell AD, Godfryd A, Buys DR, Locher JL. Does participation in home-delivered meals programs improve outcomes for older adults? Results of a systematic review. J Nutr Gerontol Geriatr. 2015;34(2):124-167. doi: 10.1080/21551197.2015.1038463. DOI: https://doi.org/10.1080/21551197.2015.1038463
Ajarma KY, Al-Faouri AF, Al Ruhaibeh MK, Almbaidien FA, Nserat RT, Al-Shawabkeh AO, et al. The risk of thyroid carcinoma in multinodular goiter compared to solitary thyroid nodules: A retrospective analysis of 600 patients. Med J Armed Forces India. 2020;76(1):23-29. doi: 10.1016/j.mjafi.2018.05.001. DOI: https://doi.org/10.1016/j.mjafi.2018.05.001
Noel KI, Abdulateef HH, Khamees NH. Pluripotency stem cell marker expression and apoptotic changes in placental tissues of normal and intra-uterine growth restriction (IUGR) babies of Iraqi mothers: A comparative study. Al-Rafidain J Med Sci. 2025;8(1):71-77. doi: 10.54133/ajms.v8i1.1642. DOI: https://doi.org/10.54133/ajms.v8i1.1642
Al-Rubai Aj, Ibraheem MM, Hameed AF, Noel KI, Eleiwi SA. Comparison of placental expression of basic fibroblast growth factor and insulin-like growth factor-1 in placentae of normal, pregnancy-induced hypertension, and preeclamptic pregnancies in Iraqi mothers. Med J Babylon. 2023;20(4):681-688. doi: 10.4103/MJBL.MJBL_40_23. DOI: https://doi.org/10.4103/MJBL.MJBL_40_23
Noel KI, Ibraheem MM, Ahmed BS, Hameed AF, Khamees NH, Akkila SS. CD133 and CD166 Expression Predicting the Possibility of Prostatic Cancer Development in Cases of BPH. Biomed Pharmacol J. 2019;12(3). Available from: https://bit.ly/2kKXP85.
Hameed AF, Noel KI, Akkila SS. Placental angiogenesis, IUGR & CMV awareness in Iraqi women. Curr Issues Pharm Med Sci. 2022;35(3):147-51. doi: 10.2478/cipms-2022-0027. DOI: https://doi.org/10.2478/cipms-2022-0027
Thompson SD, Franklyn JA, Watkinson JC, Verhaeg JM, Sheppard MC, Eggo MC. Fibroblast growth factors 1 and 2 and fibroblast growth factor receptor 1 are elevated in thyroid hyperplasia. J Clin Endocrinol Metab. 1998;83(4):1336-341. doi: 10.1210/jcem.83.4.4723. DOI: https://doi.org/10.1210/jcem.83.4.4723
Redler A, Di Rocco G, Giannotti D, Frezzotti F, Bernieri MG, Ceccarelli S, et al. Fibroblast growth factor receptor-2 expression in thyroid tumor progression: potential diagnostic application. PLoS One. 2013;8(8):e72224. doi: 10.1371/journal.pone.0072224. DOI: https://doi.org/10.1371/journal.pone.0072224
Vanderpump MPJ. The epidemiology of thyroid disease. Br Med Bull. 2011;99(1):39–51. doi: 10.1093/bmb/ldr030. DOI: https://doi.org/10.1093/bmb/ldr030
Brown EDL, Obeng-Gyasi B, Hall JE, Shekhar S. The thyroid hormone axis and female reproduction. Int J Mol Sci. 2023;24(12):9815. doi: 10.3390/ijms24129815. DOI: https://doi.org/10.3390/ijms24129815
Salman MA. Prevalence of thyroid hormones test abnormality in females at reproductive age attending Al-Batool Maternity Teaching Hospital. Diyala J Med. 2023;24(2):93-99. doi: 10.26505/djm.v24i2.1000. DOI: https://doi.org/10.26505/DJM.24027111208
Valenzano M, Giaccherino RR, Pagano L, Garberoglio S, Garberoglio R. Ultrasound assessment and sexual dimorphism of thyroid nodules: Bringing gender medicine to clinical practice. Endocrines. 2025;6(2):15. doi: 10.3390/endocrines6020015. DOI: https://doi.org/10.3390/endocrines6020015
Jaffar R, Mohanty SK, Khan A, Fischer AH. Hemosiderin laden macrophages and hemosiderin within follicular cells distinguish benign follicular lesions from follicular neoplasms. Cytojournal. 2009;6:3. doi: 10.4103/1742-6413.45193. DOI: https://doi.org/10.4103/1742-6413.45193
Beynon ME, Pinneri K. An overview of the thyroid gland and thyroid-related deaths for the forensic pathologist. Acad Forensic Pathol. 2016;6(2):217-236. doi: 10.23907/2016.024. DOI: https://doi.org/10.23907/2016.024
Melaccio A, Sgaramella LI, Pasculli A, Di Meo G, Gurrado A, Prete FP, et al. Prognostic and therapeutic role of angiogenic microenvironment in thyroid cancer. Cancers (Basel). 2021;13(11):2775. doi: 10.3390/cancers13112775. DOI: https://doi.org/10.3390/cancers13112775
Neta G, Brenner AV, Sturgis EM, Pfeiffer RM, Hutchinson AA, Aschebrook-Kilfoy B, et al. Common genetic variants related to genomic integrity and risk of papillary thyroid cancer. Carcinogenesis. 2011;32(8):1231-1237. doi: 10.1093/carcin/bgr100. DOI: https://doi.org/10.1093/carcin/bgr100
Dora SV, Rybakova MG, Alekseev DA, Krylova YS, Volkova AR, Belyakova LA. Molecular biological markers for proliferation, apoptosis, and angiogenesis in diffuse toxic goiter. Arkhiv Patologii. 2017;79(6):3-7. doi: 10.17116/patol20177963-7. DOI: https://doi.org/10.17116/patol20177963-7
Baştürk E, Kement M, Yavuzer D, Vural S, Gezen C, Gözü HI, et al. The role of insulin-like growth factor 1 in the development of benign and malignant thyroid nodules. Balkan Med J. 2012;29(2):133-138. doi: 10.5152/balkanmedj.2011.034. DOI: https://doi.org/10.5152/balkanmedj.2011.034
Nissen LJ, Cao R, Hedlund EM, Wang Z, Zhao X, Wetterskog D, et al. Angiogenic factors FGF2 and PDGF-BB synergistically promote murine tumor neovascularization and metastasis. J Clin Invest. 2007;117(10):2766-2777. doi: 10.1172/JCI32479. DOI: https://doi.org/10.1172/JCI32479
Li Y, Cha SB, Park Y, Gong BH, Jeong IY, Kim HS, et al. Evaluation of Active CASP 3and Ki-67 expression in squamous cell hyperplasia of the stomach induced by Platycodi radix water extract in Sprague-Dawley rats. J Toxicol Pathol. 2022;35(1):45-52. doi: 10.1293/tox.2021-0003. DOI: https://doi.org/10.1293/tox.2021-0003
Wen Z, Luo D, Wang S, Rong R, Evers BM, Jia L, et al. Deep learning-based H-Score quantification of immunohistochemistry-stained images. Mod Pathol. 2024;37(2):100398. doi: 10.1016/j.modpat.2023.100398. DOI: https://doi.org/10.1016/j.modpat.2023.100398
Wiedlocha A, Haugsten EM, Zakrzewska M. Roles of the FGF-FGFR signaling system in cancer development and inflammation. Cells. 2021;10(9):2231. doi: 10.3390/cells10092231. DOI: https://doi.org/10.3390/cells10092231
Boelaert K, McCabe CJ, Tannahill LA, Gittoes NJ, Holder RL, Watkinson JC, et al. Pituitary tumor transforming gene and fibroblast growth factor-2 expression: potential prognostic indicators in differentiated thyroid cancer. J Clin Endocrinol Metab. 2003;88(5):2341-2347. doi: 10.1210/jc.2002-021113. DOI: https://doi.org/10.1210/jc.2002-021113
Silva FFV, Padín-Iruegas ME, Caponio VCA, Lorenzo-Pouso AI, Saavedra-Nieves P, Chamorro-Petronacci CM, et al. Caspase 3 and cleaved caspase 3 expression in tumorogenesis and its correlations with prognosis in head and neck cancer: A systematic review and meta-analysis. Int J Mol Sci. 2022;23(19):11937. doi: 10.3390/ijms231911937. DOI: https://doi.org/10.3390/ijms231911937
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Al-Rafidain Journal of Medical Sciences ( ISSN 2789-3219 )

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Published by Al-Rafidain University College. This is an open access journal issued under the CC BY-NC-SA 4.0 license (https://creativecommons.org/licenses/by-nc-sa/4.0/).











