An Overview of Metastatic Organotropism: Mechanisms and Emerging Therapeutic Targets
DOI:
https://doi.org/10.54133/ajms.v10i2.2774Keywords:
Dormancy, Metastasis, Organotropism, Pre-metastatic niche, ReawakeningAbstract
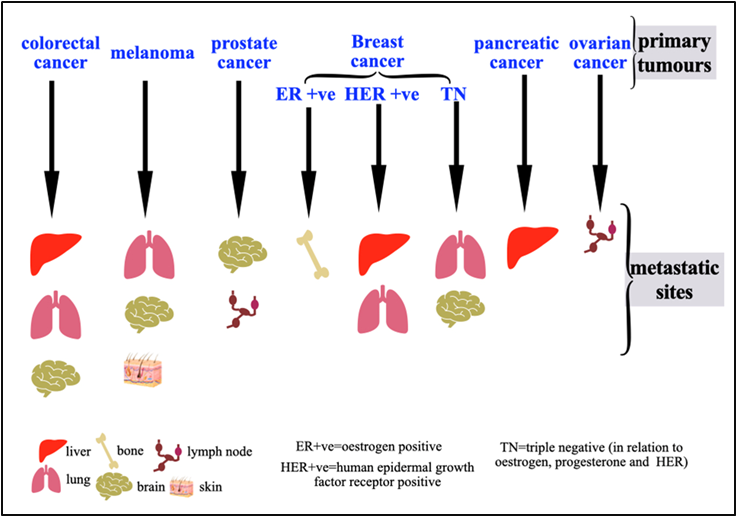
The main cause of cancer-related mortality is metastasis, which is the spread of tumor cells from their initial location to distant sites in the body. The process is not random but follows a distinct pattern of organotropism, in which certain cancers preferentially colonize distant sites. For example, estrogen-positive breast cancer may metastasize to the bones or colorectal cancer to the liver. Understanding the molecular drivers of organotropism is critical for developing effective therapies. This work combines the traditional "seed and soil" theory with modern molecular discoveries to provide a thorough overview of the current mechanistic framework underlying metastatic organotropism. We describe the multistep journey of circulating tumor cells, emphasizing key determinants such as the intrinsic properties of cancer cells, the creation of a favorable pre-metastatic niche, the interactions between disseminated cells and the microenvironment, and the role of the immune system in metastasis. We discuss organ-specific strategies aimed at disrupting the metastatic cascade, including targeting niche-forming pathways, intercepting cancer cell adhesion and modulating the immune microenvironment. Finally, we outline future challenges and opportunities, emphasizing the need for advanced models and integrative multi-omics to underscore the shifting therapeutic paradigm from late-stage generalized treatment to early targeted prevention of metastasis.
Downloads
References
Chaffer CL, Weinberg RA. A Perspective on cancer cell metastasis. Science (1979). 2011;331(6024):1559–2564. doi: 10.1126/science.1203543. DOI: https://doi.org/10.1126/science.1203543
Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19(11):1423–1437. doi: 10.1038/nm.3394. DOI: https://doi.org/10.1038/nm.3394
Lambert AW, Pattabiraman DR, Weinberg RA. Emerging biological principles of metastasis. Cell. 2017;168(4):670–691. doi: 10.1016/j.cell.2016.11.037. DOI: https://doi.org/10.1016/j.cell.2016.11.037
Alečković M, McAllister SS, Polyak K. Metastasis as a systemic disease: molecular insights and clinical implications. Biochim Biophys Rev Cancer. 2019;1872(1):89–102. doi: 10.1016/j.bbcan.2019.06.002. DOI: https://doi.org/10.1016/j.bbcan.2019.06.002
Massagué J, Ganesh K. Metastasis-initiating cells and ecosystems. Cancer Discov. 2021;11(4):971–994. doi: 10.1158/2159-8290.CD-21-0010. DOI: https://doi.org/10.1158/2159-8290.CD-21-0010
Gerstberger S, Jiang Q, Ganesh K. Metastasis. Cell. 2023;186(8):1564–1579. doi: 10.1016/j.cell.2023.03.003. DOI: https://doi.org/10.1016/j.cell.2023.03.003
Herman H, Fazakas C, Haskó J, Molnár K, Mészáros Á, Nyúl‐Tóth Á, et al. Paracellular and transcellular migration of metastatic cells through the cerebral endothelium. J Cell Mol Med. 2019;23(4):2619–2631. doi: 10.1111/jcmm.14156. DOI: https://doi.org/10.1111/jcmm.14156
Massagué J, Obenauf AC. Metastatic colonization by circulating tumour cells. Nature. 2016;529(7586):298–306. doi: 10.1038/nature17038. DOI: https://doi.org/10.1038/nature17038
Luzzi KJ, MacDonald IC, Schmidt EE, Kerkvliet N, Morris VL, Chambers AF, et al. Multistep nature of metastatic inefficiency. Am J Pathol. 1998;153(3):865–873. doi: 10.1016/s0002-9440(10)65628-3. DOI: https://doi.org/10.1016/S0002-9440(10)65628-3
Ganesh K, Massagué J. Targeting metastatic cancer. Nat Med. 2021;27(1):34–44. doi: 10.1038/s41591-020-01195-4. DOI: https://doi.org/10.1038/s41591-020-01195-4
Obenauf AC, Massagué J. Surviving at a distance: Organ-specific metastasis. Trends Cancer. 2015;1(1):76–91. doi: 10.1016/j.trecan.2015.07.009. DOI: https://doi.org/10.1016/j.trecan.2015.07.009
Li Y, Liu F, Cai Q, Deng L, Ouyang Q, Zhang XHF, et al. Invasion and metastasis in cancer: molecular insights and therapeutic targets. Signal Transduct Target Ther. 2025;10(1). doi: 10.1038/s41392-025-02148-4. DOI: https://doi.org/10.1038/s41392-025-02148-4
Gao Y, Bado I, Wang H, Zhang W, Rosen JM, Zhang XHF. Metastasis organotropism: Redefining the congenial soil. Dev Cell. 2019;49(3):375–391. doi: 10.1016/j.devcel.2019.04.012. DOI: https://doi.org/10.1016/j.devcel.2019.04.012
Nguyen DX, Bos PD, Massagué J. Metastasis: from dissemination to organ-specific colonization. Nat Rev Cancer. 2009;9(4):274–284. doi: 10.1038/nrc2622. DOI: https://doi.org/10.1038/nrc2622
Budczies J, von Winterfeld M, Klauschen F, Bockmayr M, Lennerz JK, Denkert C, et al. The landscape of metastatic progression patterns across major human cancers. Oncotarget. 2014;6(1):570–583. doi: 10.18632/oncotarget.2677. DOI: https://doi.org/10.18632/oncotarget.2677
Lu Y, Lian S, Cheng Y, Ye Y, Xie X, Fu C, et al. Circulation patterns and seed-soil compatibility factors cooperate to cause cancer organ-specific metastasis. Exp Cell Res. 2019;375(1):62–72. doi: 10.1016/j.yexcr.2018.12.015. DOI: https://doi.org/10.1016/j.yexcr.2018.12.015
Wu Q, Li J, Zhu S, Wu J, Chen C, Liu Q, et al. Breast cancer subtypes predict the preferential site of distant metastases: a SEER based study. Oncotarget. 2017;8(17):27990–27996. doi: 10.18632/oncotarget.15856. DOI: https://doi.org/10.18632/oncotarget.15856
Chen W, Hoffmann AD, Liu H, Liu X. Organotropism: new insights into molecular mechanisms of breast cancer metastasis. NPJ Precis Oncol. 2018;2(1). doi: 10.1038/s41698-018-0047-0. DOI: https://doi.org/10.1038/s41698-018-0047-0
Carrolo M, Miranda JAI, Vilhais G, Quintela A, Sousa MF, Costa DA, et al. Metastatic organotropism: a brief overview. Front Oncol. 2024;14. doi: 10.3389/fonc.2024.1358786. DOI: https://doi.org/10.3389/fonc.2024.1358786
Weiss L, Haydock K, Pickren JW, Lane WW. Organ vascularity and metastatic frequency. Am J Pathol. 1980;101(1):101–113. PMID: 7446696.
Nguyen B, Fong C, Luthra A, Smith SA, DiNatale RG, Nandakumar S, et al. Genomic characterization of metastatic patterns from prospective clinical sequencing of 25,000 patients. Cell. 2022;185(3):563-575. doi: 10.1016/j.cell.2022.01.003. DOI: https://doi.org/10.1016/j.cell.2022.01.003
Peinado H, Zhang H, Matei IR, Costa-Silva B, Hoshino A, Rodrigues G, et al. Pre-metastatic niches: organ-specific homes for metastases. Nat Rev Cancer. 2017;17(5):302–317. doi: 10.1038/nrc.2017.6. DOI: https://doi.org/10.1038/nrc.2017.6
Oskarsson T, Batlle E, Massagué J. Metastatic stem cells: Sources, niches, and vital pathways. Cell Stem Cell. 2014;14(3):306–321. doi: 10.1016/j.stem.2014.02.002. DOI: https://doi.org/10.1016/j.stem.2014.02.002
Batlle E, Clevers H. Cancer stem cells revisited. Nat Med. 2017;23(10):1124–1134. doi: 10.1038/nm.4409. DOI: https://doi.org/10.1038/nm.4409
Celià-Terrassa T, Kang Y. Metastatic niche functions and therapeutic opportunities. Nat Cell Biol. 2018;20(8):868–877. doi: 10.1038/s41556-018-0145-9. DOI: https://doi.org/10.1038/s41556-018-0145-9
Wang Y, Jia J, Wang F, Fang Y, Yang Y, Zhou Q, et al. Pre-metastatic niche: formation, characteristics and therapeutic implication. Signal Transduct Target Ther. 2024;9(1). doi: 10.1038/s41392-024-01937-7. DOI: https://doi.org/10.1038/s41392-024-01937-7
Al-Janabi I. Epithelial-mesenchymal transition and tumour progression: An overview. Al-Rafidain J Med Sci. 2023; 4:6–21. doi: 10.54133/ajms.v4i.99. DOI: https://doi.org/10.54133/ajms.v4i.99
Al-Janabi I. Manipulating cell death in cancer. Al-Rafidain J Med Sci. 2025;9(1):63–73. doi: 10.54133/ajms.v9i1.2131. DOI: https://doi.org/10.54133/ajms.v9i1.2131
Xiao G, Wang X, Xu Z, Liu Y, Jing J. Lung-specific metastasis: the coevolution of tumor cells and lung microenvironment. Mol Cancer. 2025;24(1). doi: 10.1186/s12943-025-02318-6. DOI: https://doi.org/10.1186/s12943-025-02318-6
Tao L, Huang G, Song H, Chen Y, Chen L. Cancer associated fibroblasts: An essential role in the tumor microenvironment. Oncol Lett. 2017;14(3):2611–2620. doi: 10.3892/ol.2017.6497. DOI: https://doi.org/10.3892/ol.2017.6497
Goswami D, Vestweber D. How leukocytes trigger opening and sealing of gaps in the endothelial barrier. F1000Res. 2016;5:2321. doi: 10.12688/f1000research.9185.1. DOI: https://doi.org/10.12688/f1000research.9185.1
Schuster E, Taftaf R, Reduzzi C, Albert MK, Romero-Calvo I, Liu H. Better together: circulating tumor cell clustering in metastatic cancer. Trends Cancer. 2021;7(11):1020–1032. doi: 10.1016/j.trecan.2021.07.001. DOI: https://doi.org/10.1016/j.trecan.2021.07.001
Goddard ET, Linde MH, Srivastava S, Klug G, Shabaneh TB, Iannone S, et al. Immune evasion of dormant disseminated tumor cells is due to their scarcity and can be overcome by T cell immunotherapies. Cancer Cell. 2024;42(1):119-134. doi: 10.1016/j.ccell.2023.12.011. DOI: https://doi.org/10.1016/j.ccell.2023.12.011
Harjunpää H, Llort Asens M, Guenther C, Fagerholm SC. Cell adhesion molecules and their Roles and regulation in the immune and tumor microenvironment. Front Immunol. 2019;10. doi: 10.3389/fimmu.2019.01078. DOI: https://doi.org/10.3389/fimmu.2019.01078
Loh JJ, Ma S. Hallmarks of cancer stemness. Cell Stem Cell. 2024;31(5):617–639. doi: 10.1016/j.stem.2024.04.004. DOI: https://doi.org/10.1016/j.stem.2024.04.004
Gu X, Wei S, Lv X. Circulating tumor cells: from new biological insights to clinical practice. Signal Transduct Target Ther. 2024;9(1). doi: 10.1038/s41392-024-01938-6. DOI: https://doi.org/10.1038/s41392-024-01938-6
Lin D, Shen L, Luo M, Zhang K, Li J, Yang Q, et al. Circulating tumor cells: biology and clinical significance. Signal Transduct Target Ther. 2021;6(1). doi: 10.1038/s41392-021-00817-8. DOI: https://doi.org/10.1038/s41392-021-00817-8
Nash KT, Phadke PA, Navenot JM, Hurst DR, Accavitti-Loper MA, Sztul E, et al. Requirement of KISS1 secretion for multiple organ metastasis suppression and maintenance of tumor dormancy. J Natl Cancer Institute. 2007;99(4):309–321. doi: 10.1093/jnci/djk053. DOI: https://doi.org/10.1093/jnci/djk053
Yang X, Wu JS, Li M, Zhang WL, Gao XL, Wang HF, et al. Inhibition of DEC2 is necessary for exiting cell dormancy in salivary adenoid cystic carcinoma. J Exp Clin Cancer Res. 2021;40(1):169. doi: 10.1186/s13046-021-01956-0. DOI: https://doi.org/10.1186/s13046-021-01956-0
Park SY, Nam JS. The force awakens: metastatic dormant cancer cells. Exp Mol Med. 2020;52(4):569-581. doi: 10.1038/s12276-020-0423-z. DOI: https://doi.org/10.1038/s12276-020-0423-z
Tsilimigras DI, Brodt P, Clavien PA, Muschel RJ, D’Angelica MI, Endo I, et al. Liver metastases. Nat Rev Dis Primers. 2021;7(1). doi: 10.1038/s41572-021-00261-6. DOI: https://doi.org/10.1038/s41572-021-00261-6
Chandra R, Karalis JD, Liu C, Murimwa GZ, Voth Park J, Heid CA, et al. The colorectal cancer tumor microenvironment and its impact on liver and lung metastasis. Cancers (Basel). 2021;13(24):6206. doi: 10.3390/cancers13246206. DOI: https://doi.org/10.3390/cancers13246206
Riihimäki M, Hemminki A, Sundquist J, Hemminki K. Patterns of metastasis in colon and rectal cancer. Sci Rep. 2016;6(1). doi: 10.1038/srep29765. DOI: https://doi.org/10.1038/srep29765
Domanska UM, Kruizinga RC, Nagengast WB, Timmer-Bosscha H, Huls G, de Vries EGE, et al. A review on CXCR4/CXCL12 axis in oncology: No place to hide. Eur J Cancer. 2013;49(1):219–230. doi: 10.1016/j.ejca.2012.05.005. DOI: https://doi.org/10.1016/j.ejca.2012.05.005
Mima K, Okabe H, Ishimoto T, Hayashi H, Nakagawa S, Kuroki H, et al. The expression levels of CD44v6 are correlated with the invasiveness of hepatocellular carcinoma in vitro, but do not appear to be clinically significant. Oncol Lett. 2012;3(5):1047–1051. doi: 10.3892/ol.2012.611. DOI: https://doi.org/10.3892/ol.2012.611
Lee JH, Lee SW. The roles of carcinoembryonic antigen in liver metastasis and therapeutic approaches. Gastroenterol Res Pract. 2017;2017:1–11. doi: 10.1155/2017/7521987. DOI: https://doi.org/10.1155/2017/7521987
Organ SL, Tsao MS. An overview of the c-MET signaling pathway. Ther Adv Med Oncol. 2011;3(1_suppl):S7–19. doi: 10.1177/1758834011422556. DOI: https://doi.org/10.1177/1758834011422556
Zeng ZS, Weiser MR, Kuntz E, Chen CT, Khan SA, Forslund A, et al. c-Met gene amplification is associated with advanced stage colorectal cancer and liver metastases. Cancer Lett. 2008;265(2):258–269. doi: 10.1016/j.canlet.2008.02.049. DOI: https://doi.org/10.1016/j.canlet.2008.02.049
Takeda M, Yoshida S, Inoue T, Sekido Y, Hata T, Hamabe A, et al. The role of KRAS mutations in colorectal cancer: Biological insights, clinical implications, and future therapeutic perspectives. Cancers (Basel). 2025;17(3):428. doi: 10.3390/cancers17030428. DOI: https://doi.org/10.3390/cancers17030428
Said R, Hong DS, Warneke CL, Lee JJ, Wheler JJ, Janku F, et al. P53 Mutations in advanced cancers: Clinical characteristics, outcomes, and correlation between progression-free survival and bevacizumab-containing therapy. Oncotarget. 2013;4(5):705–714. doi: 10.18632/oncotarget.974. DOI: https://doi.org/10.18632/oncotarget.974
Kimbung S, Johansson I, Danielsson A, Veerla S, Egyhazi Brage S, Frostvik Stolt M, et al. Transcriptional profiling of breast cancer metastases identifies liver metastasis–selective genes associated with adverse outcome in luminal A primary breast cancer. Clin Cancer Res. 2016;22(1):146–157. doi: 10.1158/1078-0432.CCR-15-0487. DOI: https://doi.org/10.1158/1078-0432.CCR-15-0487
Harada K, Carr SM, Shrestha A, La Thangue NB. Citrullination and the protein code: crosstalk between post-translational modifications in cancer. Philos Trans R Soc Lond B Biol Sci. 2023;378(1890):20220243. doi: 10.1098/rstb.2022.0243. DOI: https://doi.org/10.1098/rstb.2022.0243
Tabariès S, Dupuy F, Dong Z, Monast A, Annis MG, Spicer J, et al. Claudin-2 promotes breast cancer liver metastasis by facilitating tumor cell interactions with hepatocytes. Mol Cell Biol. 2012;32(15):2979–2991. doi: 10.1128/MCB.00299-12. DOI: https://doi.org/10.1128/MCB.00299-12
Tian Z, Hou X, Liu W, Han Z, Wei L. Macrophages and hepatocellular carcinoma. Cell Biosci. 2019;9:79. doi: 10.1186/s13578-019-0342-7. DOI: https://doi.org/10.1186/s13578-019-0342-7
Enns A, Korb T, Schlüter K, Gassmann P, Spiegel HU, Senninger N, et al. αvβ5-Integrins mediate early steps of metastasis formation. Eur J Cancer. 2005;41(7):1065–1072. doi: 10.1016/j.ejca.2004.12.031. DOI: https://doi.org/10.1016/j.ejca.2004.12.031
Zhang T, Yang Z, Kusumanchi P, Han S, Liangpunsakul S. Critical role of microRNA-21 in the pathogenesis of liver diseases. Front Med (Lausanne). 2020;7. doi: 10.3389/fmed.2020.00007. DOI: https://doi.org/10.3389/fmed.2020.00007
Hu J, Xu Y, Hao J, Wang S, Li C, Meng S. MiR-122 in hepatic function and liver diseases. Protein Cell. 2012;3(5):364–371. doi: 10.1007/s13238-012-2036-3. DOI: https://doi.org/10.1007/s13238-012-2036-3
Costa-Silva B, Aiello NM, Ocean AJ, Singh S, Zhang H, Thakur BK, et al. Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nat Cell Biol. 2015;17(6):816–826. doi: 10.1038/ncb3169. DOI: https://doi.org/10.1038/ncb3169
Van den Eynden GG, Majeed AW, Illemann M, Vermeulen PB, Bird NC, Høyer-Hansen G, et al. The multifaceted role of the microenvironment in liver metastasis: Biology and clinical implications. Cancer Res. 2013;73(7):2031–2043. doi: 10.1158/0008-5472.CAN-12-3931. DOI: https://doi.org/10.1158/0008-5472.CAN-12-3931
Medeiros B, Allan AL. Molecular mechanisms of breast cancer metastasis to the lung: Clinical and experimental perspectives. Int J Mol Sci. 2019;20(9):2272. doi: 10.3390/ijms20092272. DOI: https://doi.org/10.3390/ijms20092272
Minn AJ, Gupta GP, Siegel PM, Bos PD, Shu W, Giri DD, et al. Genes that mediate breast cancer metastasis to lung. Nature. 2005;436(7050):518–524. doi: 10.1038/nature03799. DOI: https://doi.org/10.1038/nature03799
Hou J, Zhang Y, Zhu Z. Gene heterogeneity in metastasis of colorectal cancer to the lung. Semin Cell Dev Biol. 2017;64:58-64. doi: 10.1016/j.semcdb.2016.08.034. DOI: https://doi.org/10.1016/j.semcdb.2016.08.034
Mulholland DJ, Kobayashi N, Ruscetti M, Zhi A, Tran LM, Huang J, et al. Pten loss and RAS/MAPK activation cooperate to promote EMT and metastasis initiated from prostate cancer stem/progenitor cells. Cancer Res. 2012;72(7):1878–1889. doi: 10.1158/0008-5472.CAN-11-3132. DOI: https://doi.org/10.1158/0008-5472.CAN-11-3132
Appert-Collin A, Hubert P, Crémel G, Bennasroune A. Role of ErbB receptors in cancer cell migration and invasion. Front Pharmacol. 2015;6. doi: 10.3389/fphar.2015.00283. DOI: https://doi.org/10.3389/fphar.2015.00283
Zlotnik A, Burkhardt AM, Homey B. Homeostatic chemokine receptors and organ-specific metastasis. Nat Rev Immunol. 2011;11(9):597–606. doi: 10.1038/nri3049. DOI: https://doi.org/10.1038/nri3049
Li F, Xu X, Geng J, Wan X, Dai H. The autocrine CXCR4/CXCL12 axis contributes to lung fibrosis through modulation of lung fibroblast activity. Exp Ther Med. 2020. doi: 10.3892/etm.2020.8433. DOI: https://doi.org/10.3892/etm.2020.8433
Zhu D, Cheng CF, Pauli BU. Blocking of lung endothelial cell adhesion molecule-1 (Lu-ECAM-1) inhibits murine melanoma lung metastasis. J Clin Invest. 1992;89(6):1718–1724. doi: 10.1172/JCI115773. DOI: https://doi.org/10.1172/JCI115773
Kang SA, Hasan N, Mann AP, Zheng W, Zhao L, Morris L, et al. Blocking the adhesion cascade at the premetastatic niche for prevention of breast cancer metastasis. Mol Ther. 2015;23(6):1044–1054. doi: 10.1038/mt.2015.45. DOI: https://doi.org/10.1038/mt.2015.45
Huang R, Rofstad EK. Integrins as therapeutic targets in the organ-specific metastasis of human malignant melanoma. J Exp Clin Cancer Res. 2018;37(1). doi: 10.1186/s13046-018-0763-x. DOI: https://doi.org/10.1186/s13046-018-0763-x
Wu S, Zheng Q, Xing X, Dong Y, Wang Y, You Y, et al. Matrix stiffness-upregulated LOXL2 promotes fibronectin production, MMP9 and CXCL12 expression and BMDCs recruitment to assist pre-metastatic niche formation. J Exp Clin Cancer Res. 2018;37(1). doi: 10.1186/s13046-018-0761-z. DOI: https://doi.org/10.1186/s13046-018-0761-z
Massagué J, Sheppard D. TGF-β signaling in health and disease. Cell. 2023;186(19):4007–4037. doi: 10.1016/j.cell.2023.07.036. DOI: https://doi.org/10.1016/j.cell.2023.07.036
Makinen T. Isolated lymphatic endothelial cells transduce growth, survival and migratory signals via the VEGF-C/D receptor VEGFR-3. EMBO J. 2001;20(17):4762–4773. doi: 10.1093/emboj/20.17.4762. DOI: https://doi.org/10.1093/emboj/20.17.4762
Ji H, Hu C, Yang X, Liu Y, Ji G, Ge S, et al. Lymph node metastasis in cancer progression: molecular mechanisms, clinical significance and therapeutic interventions. Signal Transduct Target Ther. 2023;8(1). doi: 10.1038/s41392-023-01576-4. DOI: https://doi.org/10.1038/s41392-023-01576-4
Guan X. Cancer metastases: challenges and opportunities. Acta Pharm Sin B. 2015;5(5):402–418. doi: 10.1016/j.apsb.2015.07.005. DOI: https://doi.org/10.1016/j.apsb.2015.07.005
Woo HY, Rhee H, Yoo JE, Kim SH, Choi GH, Kim DY, et al. Lung and lymph node metastases from hepatocellular carcinoma: Comparison of pathological aspects. Liver Int. 2021;42(1):199–209. doi: 10.1111/liv.15051. DOI: https://doi.org/10.1111/liv.15051
Devisetti N, Shah P, Liu FC. From mechanisms to treatment: A comprehensive view of lymphatic metastasis in cancer. Lymphatics. 2025;3(2):12. doi: 10.3390/lymphatics3020012. DOI: https://doi.org/10.3390/lymphatics3020012
Follain G, Herrmann D, Harlepp S, Hyenne V, Osmani N, Warren SC, et al. Fluids and their mechanics in tumour transit: shaping metastasis. Nat Rev Cancer. 2019;20(2):107–124. doi: 10.1038/s41568-019-0221-x. DOI: https://doi.org/10.1038/s41568-019-0221-x
Swartz MA, Lund AW. Lymphatic and interstitial flow in the tumour microenvironment: linking mechanobiology with immunity. Nat Rev Cancer. 2012;12(3):210–219. doi: 10.1038/nrc3186. DOI: https://doi.org/10.1038/nrc3186
Issa A, Le TX, Shoushtari AN, Shields JD, Swartz MA. Vascular endothelial growth factor-C and C-C chemokine receptor 7 in tumor cell–lymphatic cross-talk promote invasive phenotype. Cancer Res. 2008;69(1):349–357. doi: 10.1158/0008-5472.CAN-08-1875. DOI: https://doi.org/10.1158/0008-5472.CAN-08-1875
Cornelison RC, Brennan CE, Kingsmore KM, Munson JM. Convective forces increase CXCR4-dependent glioblastoma cell invasion in GL261 murine model. Sci Rep. 2018;8(1). doi: 10.1038/s41598-018-35141-9. DOI: https://doi.org/10.1038/s41598-018-35141-9
Dogan NU, Dogan S, Favero G, Köhler C, Dursun P. The basics of sentinel lymph node biopsy: Anatomical and pathophysiological considerations and clinical aspects. J Oncol. 2019;2019:1–10. doi: 10.1155/2019/3415630. DOI: https://doi.org/10.1155/2019/3415630
Wang M, Xia F, Wei Y, Wei X. Molecular mechanisms and clinical management of cancer bone metastasis. Bone Res. 2020;8(1). doi: 10.1038/s41413-020-00105-1. DOI: https://doi.org/10.1038/s41413-020-00105-1
Wang J, Loberg R, Taichman RS. The pivotal role of CXCL12 (SDF-1)/CXCR4 axis in bone metastasis. Cancer Metastasis Rev. 2006;25(4):573–587. doi: 10.1007/s10555-006-9019-x. DOI: https://doi.org/10.1007/s10555-006-9019-x
Suva LJ, Washam C, Nicholas RW, Griffin RJ. Bone metastasis: mechanisms and therapeutic opportunities. Nat Rev Endocrinol. 2011;7(4):208–218. doi: 10.1038/nrendo.2010.227. DOI: https://doi.org/10.1038/nrendo.2010.227
Roodman GD, Dougall WC. RANK ligand as a therapeutic target for bone metastases and multiple myeloma. Cancer Treat Rev. 2008;34(1):92–101. doi: 10.1016/j.ctrv.2007.09.002. DOI: https://doi.org/10.1016/j.ctrv.2007.09.002
Arnett T. Regulation of bone cell function by acid–base balance. Proc Nutr Soc. 2003;62(2):511–520. doi: 10.1079/pns2003268. DOI: https://doi.org/10.1079/PNS2003268
Campbell BK, Gao Z, Corcoran NM, Stylli SS, Hovens CM. Molecular mechanisms driving the formation of brain metastases. Cancers (Basel). 2022;14(19):4963. doi: 10.3390/cancers14194963. DOI: https://doi.org/10.3390/cancers14194963
Alsabbagh R, Ahmed M, Alqudah MAY, Hamoudi R, Harati R. Insights into the molecular mechanisms mediating extravasation in brain metastasis of breast cancer, melanoma, and lung cancer. Cancers (Basel). 2023;15(8):2258. doi: 10.3390/cancers15082258. DOI: https://doi.org/10.3390/cancers15082258
Liang Z, Mo Y, Zhang Y, Yu Y, Ji Y. Molecular mechanisms and signaling pathways related to brain metastasis in breast cancer. Front Pharmacol. 2025;16. doi: 10.3389/fphar.2025.1585668. DOI: https://doi.org/10.3389/fphar.2025.1585668
Osaid Z, Haider M, Hamoudi R, Harati R. Exosomes interactions with the blood–brain barrier: Implications for cerebral disorders and therapeutics. Int J Mol Sci. 2023;24(21):15635. doi: 10.3390/ijms242115635. DOI: https://doi.org/10.3390/ijms242115635
Adler O, Zait Y, Cohen N, Blazquez R, Doron H, Monteran L, et al. Reciprocal interactions between innate immune cells and astrocytes facilitate neuroinflammation and brain metastasis via lipocalin-2. Nat Cancer. 2023;4(3):401–418. doi: 10.1038/s43018-023-00519-w. DOI: https://doi.org/10.1038/s43018-023-00519-w
Jacome MA, Wu Q, Chen J, Mohamed ZS, Mokhtari S, Piña Y, et al. Molecular underpinnings of brain metastases. Int J Mol Sci. 2025;26(5):2307. doi: 10.3390/ijms26052307. DOI: https://doi.org/10.3390/ijms26052307
Chen Q, Boire A, Jin X, Valiente M, Er EE, Lopez-Soto A, et al. Carcinoma–astrocyte gap junctions promote brain metastasis by cGAMP transfer. Nature. 2016;533(7604):493–498. doi: 10.1038/nature18268. DOI: https://doi.org/10.1038/nature18268
Bautista J, López-Cortés A. Deciphering organotropism reveals therapeutic targets in metastasis. Front Cell Dev Biol. 2025;13. doi: 10.3389/fcell.2025.1677481. DOI: https://doi.org/10.3389/fcell.2025.1677481
Nussinov R, Yavuz BR, Jang H. Molecular principles underlying aggressive cancers. Signal Transduct Target Ther. 2025;10(1). doi: 10.1038/s41392-025-02129-7. DOI: https://doi.org/10.1038/s41392-025-02129-7
Han QF, Li WJ, Hu KS, Gao J, Zhai WL, Yang JH, et al. Exosome biogenesis: machinery, regulation, and therapeutic implications in cancer. Mol Cancer. 2022;21(1). doi: 10.1186/s12943-022-01671-0. DOI: https://doi.org/10.1186/s12943-022-01671-0
Zheng Y, Tu C, Zhang J, Wang J. Inhibition of multiple myeloma‑derived exosomes uptake suppresses the functional response in bone marrow stromal cell. Int J Oncol. 2019. doi: 10.3892/ijo.2019.4685. DOI: https://doi.org/10.3892/ijo.2019.4685
Hirosawa KM, Sato Y, Kasai RS, Yamaguchi E, Komura N, Ando H, et al. Uptake of small extracellular vesicles by recipient cells is facilitated by paracrine adhesion signaling. Nat Commun. 2025;16(1). doi: 10.1038/s41467-025-57617-9. DOI: https://doi.org/10.1038/s41467-025-57617-9
Xu WW, Li B, Lam AKY, Tsao SW, Law SYK, Chan KW, et al. Targeting VEGFR1- and VEGFR2-expressing non-tumor cells is essential for esophageal cancer therapy. Oncotarget. 2014;6(3):1790–805. doi: 10.18632/oncotarget.2781. DOI: https://doi.org/10.18632/oncotarget.2781
Mahaki H, Nobari S, Tanzadehpanah H, Babaeizad A, Kazemzadeh G, Mehrabzadeh M, et al. Targeting VEGF signaling for tumor microenvironment remodeling and metastasis inhibition: Therapeutic strategies and insights. Biomed Pharmacother. 2025;186:118023. doi: 10.1016/j.biopha.2025.118023. DOI: https://doi.org/10.1016/j.biopha.2025.118023
Aktary Z, Alaee M, Pasdar M. Beyond cell-cell adhesion: Plakoglobin and the regulation of tumorigenesis and metastasis. Oncotarget. 2017;8(19):32270–32291. doi: 10.18632/oncotarget.15650. DOI: https://doi.org/10.18632/oncotarget.15650
Kucuk M, Kaya M, Kalayci R, Cimen V, Kudat H, Arican N, et al. Effects of losartan on the blood–brain barrier permeability in long-term nitric oxide blockade-induced hypertensive rats. Life Sci. 2002;71(8):937–946. doi: 10.1016/s0024-3205(02)01772-1. DOI: https://doi.org/10.1016/S0024-3205(02)01772-1
Lu J, Hu D, Zhang Y, Ma C, Shen L, Shuai B. Current comprehensive understanding of denosumab (the RANKL neutralizing antibody) in the treatment of bone metastasis of malignant tumors, including pharmacological mechanism and clinical trials. Front Oncol. 2023;13. doi: 10.3389/fonc.2023.1133828. DOI: https://doi.org/10.3389/fonc.2023.1133828
Kane JF, Johnson RW. Re-evaluating the role of PTHrP in breast cancer. Cancers (Basel). 2023;15(10):2670. doi: 10.3390/cancers15102670. DOI: https://doi.org/10.3390/cancers15102670
Yang H, Shi T, Dong J, Zhang T, Li Y, Guo Y, et al. Structural insights into the substrate transport mechanism of the amino acid transporter complex. J Biol Chem. 2025;301(9):110569. doi: 10.1016/j.jbc.2025.110569. DOI: https://doi.org/10.1016/j.jbc.2025.110569
Gu L, Zhu Y, Lee M, Nguyen A, Ryujin NT, Huang JY, et al. Angiotensin II receptor inhibition ameliorates liver fibrosis and enhances hepatocellular carcinoma infiltration by effector T cells. Proc Natl Acad Sci. 2023;120(19). doi: 10.1073/pnas.2300706120. DOI: https://doi.org/10.1073/pnas.2300706120
Emanuelli A, Souleyreau W, Chouleur T, Boeckx B, Pobiedonoscew Y, Cooley L, et al. Targeting the IL34-CSF1R axis improves metastatic renal cell carcinoma therapy outcome via immune-vascular crosstalk regulation. iScience. 2025;28(6):112752. doi: 10.1016/j.isci.2025.112752. DOI: https://doi.org/10.1016/j.isci.2025.112752
Summers MA, McDonald MM, Croucher PI. Cancer cell dormancy in metastasis. Cold Spring Harb Perspect Med. 2019;10(4):a037556. doi: 10.1101/cshperspect.a037556. DOI: https://doi.org/10.1101/cshperspect.a037556
Ghajar CM. Metastasis prevention by targeting the dormant niche. Nat Rev Cancer. 2015;15(4):238–247. doi: 10.1038/nrc3910. DOI: https://doi.org/10.1038/nrc3910
Hu J, Sánchez-Rivera FJ, Wang Z, Johnson GN, Ho YJ, Ganesh K, et al. STING inhibits the reactivation of dormant metastasis in lung adenocarcinoma. Nature. 2023;616(7958):806–813. doi: 10.1038/s41586-023-05880-5. DOI: https://doi.org/10.1038/s41586-023-05880-5
Kosaka N, Iguchi H, Hagiwara K, Yoshioka Y, Takeshita F, Ochiya T. Neutral sphingomyelinase 2 (nSMase2)-dependent exosomal transfer of angiogenic microRNAs regulate cancer cell metastasis. J Biol Chem. 2013;288(15):10849–10859. doi: 10.1074/jbc.M112.446831. DOI: https://doi.org/10.1074/jbc.M112.446831
Nishida-Aoki N, Tominaga N, Takeshita F, Sonoda H, Yoshioka Y, Ochiya T. Disruption of circulating extracellular vesicles as a novel therapeutic strategy against cancer metastasis. Mol Ther. 2017;25(1):181–191. doi: 10.1016/j.ymthe.2016.10.009. DOI: https://doi.org/10.1016/j.ymthe.2016.10.009
Aceto N, Bardia A, Miyamoto DT, Donaldson MC, Wittner BS, Spencer JA, et al. Circulating tumor cell clusters are oligoclonal precursors of breast cancer metastasis. Cell. 2014;158(5):1110–1122. doi: 10.1016/j.cell.2014.07.013. DOI: https://doi.org/10.1016/j.cell.2014.07.013
Taipaleenmäki H, Drake MT. Bone anabolic therapy in breast cancer: is there a role for parathyroid hormone or parathyroid hormone-related protein? JBMR Plus. 2025;9(9). doi: 10.1093/jbmrpl/ziaf108. DOI: https://doi.org/10.1093/jbmrpl/ziaf108
Yang Z, Yang L, Zhang J, Qian C, Zhao Y. AS602801 treatment suppresses breast cancer metastasis to the brain by interfering with gap‐junction communication by regulating Cx43 expression. Drug Dev Res. 2023;85(1). doi: 10.1002/ddr.22124. DOI: https://doi.org/10.1002/ddr.22124
Zhang Z, Lin J, Yang L, Li Y. Osimertinib inhibits brain metastases and improves long-term survival in a patient with advanced squamous cell lung cancer: a case report and literatures review. Front Oncol. 2023;13. doi: 10.3389/fonc.2023.1188772. DOI: https://doi.org/10.3389/fonc.2023.1188772
Parker AL, Benguigui M, Fornetti J, Goddard E, Lucotti S, Insua-Rodríguez J, et al. Current challenges in metastasis research and future innovation for clinical translation. Clin Exp Metastasis. 2022;39(2):263–277. doi: 10.1007/s10585-021-10144-5. DOI: https://doi.org/10.1007/s10585-021-10144-5
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Al-Rafidain Journal of Medical Sciences ( ISSN 2789-3219 )

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Published by Al-Rafidain University College. This is an open access journal issued under the CC BY-NC-SA 4.0 license (https://creativecommons.org/licenses/by-nc-sa/4.0/).











