Griseofulvin Nanosuspension: A Quality by Design Approach to Enhance the Dissolution Performance
DOI:
https://doi.org/10.54133/ajms.v10i1.2693Keywords:
Box–Behnken design, Cryoprotectant, Lyophilization , Nanosuspension , Particle size, StabilityAbstract
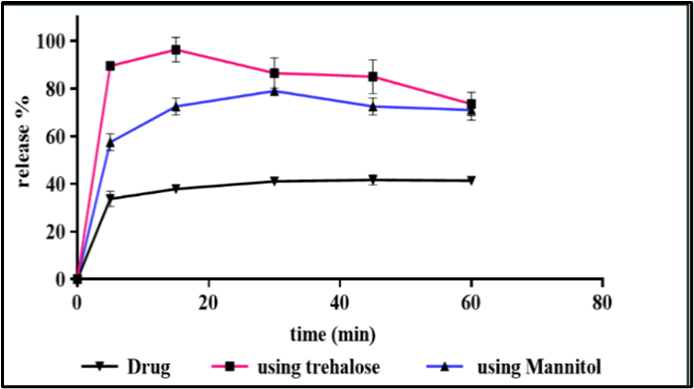
Background: Griseofulvin (GF) is a BCS class II antifungal drug characterized by poor aqueous solubility with low and variable oral bioavailability, which affects its therapeutic performance. Objective: This study aimed to optimize a stable griseofulvin nanosuspension (NS) using an ultrasonication-assisted antisolvent precipitation method combined with a Quality by Design (QbD) approach. Methods: Preliminary stabilizer screening identified polyvinylpyrrolidone K30 (PVP) and sodium lauryl sulfate (SLS) as the most effective electrosteric stabilizer system. Box–Behnken design was used to study the effects of surfactant and polymer percent, in addition to sonication time, on particle size (PS) and polydispersity index (PDI). The optimized nanosuspension was lyophilized using trehalose or mannitol. as cryoprotectants. Physicochemical characterization was performed using particle size analysis, zeta potential, PXRD, DSC, and FTIR, while solubility, dissolution, and stability studies were also conducted. Results: The optimized formulation exhibited 264±32.7 nm as PS, a PDI of 0.246±0.02, an entrapment efficiency of 91±3.44%, and a zeta potential of −28.7 mV, indicating good colloidal stability. Trehalose provided superior redispersibility and demonstrated significantly enhanced solubility and rapid dissolution, achieving 96% drug release within 15 min, compared with mannitol. Solid-state characterization confirmed a marked reduction in crystallinity and partial amorphization of GF. Stability studies confirmed acceptable physicochemical stability under accelerated storage conditions. Conclusions: Overall, the developed griseofulvin nanosuspension represents a promising strategy for improving the solubility and dissolution behavior of poorly water-soluble drugs
Downloads
References
Dhudum R, Ganeshpurkar A, Pawar A. Revolutionizing drug discovery: A comprehensive review of AI applications. Drugs Drug Candidates. 2024;3:1:148-171. doi: 10.3390/ddc3010009. DOI: https://doi.org/10.3390/ddc3010009
Truzzi F, Tibaldi C, Zhang Y, Dinelli G, D′ Amen E. An overview on dietary polyphenols and their biopharmaceutical classification system BCS. Int J Mol Sci. 2021;22:5514. doi: 10.3390/ijms22115514. DOI: https://doi.org/10.3390/ijms22115514
Bhalani DV, Nutan B, Kumar A, Singh Chandel AK. Bioavailability enhancement techniques for poorly aqueous soluble drugs and therapeutics. Biomedicines. 2022;10:2055. doi: 10.3390/biomedicines10092055. DOI: https://doi.org/10.3390/biomedicines10092055
Jacob S, Kather FS, Boddu SHS, Attimarad M, Nair AB. Nanosuspension innovations: Expanding horizons in drug delivery techniques. Pharmaceutics. 2025;17:136. doi: 10.3390/pharmaceutics17010136. DOI: https://doi.org/10.3390/pharmaceutics17010136
Chavhan R. Nanosuspensions: Enhancing drug bioavailability through nanonization. Ann Pharm Fr. 2025;83:2:251-271. doi: 10.1016/j.pharma.2024.06.003. DOI: https://doi.org/10.1016/j.pharma.2024.06.003
Noyes AA, Whitney WR. The rate of solution of solid substances in their own solutions. J Am Chem Soc. 1897;19:930-934. doi: 10.1021/ja02086a003. DOI: https://doi.org/10.1021/ja02086a003
Nguyen HX, Le NY, Nguyen CN. Quality by design optimization of formulation variables and process parameters for enhanced transdermal delivery of nanosuspension. Drug Del Transl Res. 2025;15:2220-2251. doi: 10.1007/s13346-024-01733-4. DOI: https://doi.org/10.1007/s13346-024-01733-4
USP. Griseofulvin United States Pharmacopeia 43 – National Formulary 38. Rockville, MD: United States Pharmacopeial Convention; 2020. pp. 3008-11.
Aris P, Wei Y, Mohamadzadeh M, Xia X. Griseofulvin: An updated overview of old and current knowledge. Molecules. 2022;27:7034. doi: 10.3390/molecules27207034. DOI: https://doi.org/10.3390/molecules27207034
Kumar R, Siril PF. Controlling the size and morphology of griseofulvin nanoparticles using polymeric stabilizers by evaporation-assisted solvent–antisolvent interaction method. J Nanopart Res. 2015;17:256. doi: 10.1007/s11051-015-3066-6. DOI: https://doi.org/10.1007/s11051-015-3066-6
Li M, Alvarez P, Orbe P, Bilgili E. Multi-faceted characterization of wet-milled griseofulvin nanosuspensions for elucidation of aggregation state and stabilization mechanisms. AAPS PharmSciTech. 2018;19:1789-801. doi: 10.1208/s12249-018-0993-4. DOI: https://doi.org/10.1208/s12249-018-0993-4
Sigfridsson K, Rydberg H, Strimfors M. Nano- and microcrystals of griseofulvin subcutaneously administered to rats resulted in improved bioavailability and sustained release. Drug Dev Ind Pharm. 2019;45:1477-1486. doi: 10.1080/03639045.2019.1628769. DOI: https://doi.org/10.1080/03639045.2019.1628769
Elshafeey AH, El-Dahmy RM. Formulation and development of oral fast-dissolving films loaded with nanosuspension to augment paroxetine bioavailability. Pharmaceutics. 2021;13:1869. doi: 10.3390/pharmaceutics13111869. DOI: https://doi.org/10.3390/pharmaceutics13111869
Abbas AF, Ashoor JA, Alamir HTA. Preparation and characterization of fluocinolone acetonide as nanosuspension based hydrogel for topical skin administration. Al Mustansiriyah J Pharm Sci. 2026;25:682-696. doi: 10.32947/ajps.v25i5.1201. DOI: https://doi.org/10.32947/ajps.v25i5.1201
Ghose D, Patra CN, Ravi Kumar BVV, Swain S, Jena BR, Choudhury P, et al. QbD-based formulation optimization and characterization of polymeric nanoparticles of cinacalcet hydrochloride. Turk J Pharm Sci. 2021;18:452-464. doi: 10.4274/tjps.galenos.2020.08522. DOI: https://doi.org/10.4274/tjps.galenos.2020.08522
Abd-Alhammid SN, Kadhum RW. Process factors affecting the preparation and characterization of dutasteride nanosuspension. Iraqi J Pharm Sci. 2025;34:35-48. doi: 10.31351/vol34iss1pp35-48.
Mohammed-Kadhum MF, Hameed GS. Development and characterization of furosemide-loaded binary amorphous solid dispersion. Pharmacia. 2025;72:1-19. doi: 10.3897/pharmacia.72.e156784. DOI: https://doi.org/10.3897/pharmacia.72.e156784
Mohammed AA, Abd Alhammid SN. Preparation, in vitro evaluation and characterization studies of clozapine nanosuspension. Iraqi J Pharm Sci. 2024;33:336-348. doi: 10.31351/vol33iss4SIpp336-348. DOI: https://doi.org/10.31351/vol33iss(4SI)pp336-348
Gülbağ Pınar S, Pezik E, Mutlu Ağardan B, Çelebi N. Development of cyclosporine A nanosuspension: cytotoxicity and permeability on Caco-2 cell lines. Pharm Dev Technol. 2022;27:52-62. doi: 10.1080/10837450.2021.2020817. DOI: https://doi.org/10.1080/10837450.2021.2020817
Alwan RM, Rajab NA. Nanosuspensions of selexipag: formulation, characterization, and in vitro evaluation. Iraqi J Pharm Sci. 2021;30:144-153. doi: 10.31351/vol30iss1pp144-153. DOI: https://doi.org/10.31351/vol30iss1pp144-153
Hussein AM, Hameed G, Aziz FM. Improve the solubility of cefpodoxime proxetil by amorphous solid dispersion technique. J Res Pharm. 2025;29:1437-1450. doi: 10.12991/jrespharm.1734450. DOI: https://doi.org/10.12991/jrespharm.1734450
Thorat S, Singh M, Tare M. Bioavailability enhancement of abiraterone acetate through quercetin-loaded nanoparticles. Palest Med Pharm J. 2025;11:3. doi: 10.59049/2790-0231.11.3.2502. DOI: https://doi.org/10.59049/2790-0231.11.3.2502
Maded ZK, Sfar S, Taqa GAA, Lassoued MA, Ben Hadj Ayed O, et al. Development and optimization of dipyridamole- and roflumilast-loaded nanoemulsion and nanoemulgel. Pharmaceuticals. 2024;17:6. doi: 10.3390/ph17060803. DOI: https://doi.org/10.3390/ph17060803
Porwal O. Box-Behnken design-based formulation optimization and characterization of spray dried rutin loaded nanosuspension. South Afr J Bot. 2022;149:807-815. doi: 10.1016/j.sajb.2022.04.028. DOI: https://doi.org/10.1016/j.sajb.2022.04.028
Naama NA, Hameed GS, Hanna DB, Mahdi ZH. Formulation of cefdinir ternary solid dispersion and stability study under harsh conditions. Al Mustansiriyah J Pharm Sci. 2025;25:27-48. doi: 10.32947/ajps.v25i1.1106. DOI: https://doi.org/10.32947/ajps.v25i1.1106
Alhagiesa AW, Ghareeb MM. The formulation and characterization of nimodipine nanoparticles for the enhancement of solubility and dissolution rate. Iraqi J Pharm Sci. 2021;30:143-152. doi: 10.31351/vol30iss2pp143-152. DOI: https://doi.org/10.31351/vol30iss2pp143-152
Leung DH. Development of nanosuspension formulations compatible with inkjet printing. Pharmaceutics. 2022;14:2. doi: 10.3390/pharmaceutics14020449. DOI: https://doi.org/10.3390/pharmaceutics14020449
Qureshia MJ, Phina FF, Patrob S. Enhanced solubility and dissolution rate of clopidogrel by nanosuspension. J Appl Pharm Sci. 2017;7:106-113. doi: 10.7324/JAPS.2017.70213. DOI: https://doi.org/10.7324/JAPS.2017.70213
Patel J, Dhingani A, Garala K, Raval M, Sheth N. Quality by design approach for oral bioavailability enhancement of Irbesartan. Drug Deliv. 2014;21:412-435. doi: 10.3109/10717544.2013.853709. DOI: https://doi.org/10.3109/10717544.2013.853709
Sreeharsha N, Prasanthi S, Rao GSNK, Gajula LR, Biradar N, Goudanavar P, et al. Formulation optimization of chitosan surface coated solid lipid nanoparticles of griseofulvin. Eur J Pharm Sci. 2025;204:106951. doi: 10.1016/j.ejps.2024.106951. DOI: https://doi.org/10.1016/j.ejps.2024.106951
Galipeau K, Socki M, Socia A, Harmon PA. Incomplete loading of sodium lauryl sulfate and fasted state simulated intestinal fluid micelles. J Pharm Sci. 2018;107:156-169. doi: 10.1016/j.xphs.2017.06.006. DOI: https://doi.org/10.1016/j.xphs.2017.06.006
Che Mohamed Hussein SN, Amir Z, Jan BM, Khalil M, Azizi A. Colloidal stability of CA, SDS and PVA coated iron oxide nanoparticles IONPs. Polymers. 2022;14:4787. doi: 10.3390/polym14214787. DOI: https://doi.org/10.3390/polym14214787
Karakucuk A, Celebi N. Investigation of formulation and process parameters of wet media milling to develop etodolac nanosuspensions. Pharm Res. 2020;37:111. doi: 10.1007/s11095-020-02815-x. DOI: https://doi.org/10.1007/s11095-020-02815-x
Zhang Y, Li B, Liu J, Han D, Rohani S, Gao Z, et al. Inhibition of crystal nucleation and growth: A review. Cryst Growth Des. 2024;24:2645-2665. doi: 10.1021/acs.cgd.3c01345. DOI: https://doi.org/10.1021/acs.cgd.3c01345
Siddique M, Rashid R, Ali A. Fundamentals of acoustic cavitation, ultrasound-assisted processes, and sonochemistry. Modeling and Simulation of Sono-Processes. Elsevier; 2025. p. 3-17. DOI: https://doi.org/10.1016/B978-0-443-23651-8.00001-2
Gokce Y, Cengiz B, Yildiz N, Calimli A, Aktas Z. Ultrasonication of chitosan nanoparticle suspension: Influence on particle size. Colloids and Surfaces A. 2014;462:75-81. doi: 10.1016/j.colsurfa.2014.08.028. DOI: https://doi.org/10.1016/j.colsurfa.2014.08.028
Sharma C, Desai MA, Patel SR. Effect of surfactants and polymers on morphology and particle size of telmisartan. Chem Papers. 2019;73:1685-1694. doi: 10.1007/s11696-019-00720-1. DOI: https://doi.org/10.1007/s11696-019-00720-1
Prasad R, Dalvi SV. Understanding morphological evolution of griseofulvin particles. Cryst Growth Des. 2019;19:5836-5849. doi: 10.1021/ACS.CGD.9B00859. DOI: https://doi.org/10.1021/acs.cgd.9b00859
Sabnis SS, Singh SD, Gogate PR. Improvements in azithromycin recrystallization using ultrasound. Ultrason Sonochem. 2022;83:105922. doi: 10.1016/j.ultsonch.2022.105922. DOI: https://doi.org/10.1016/j.ultsonch.2022.105922
Khan A, Naquvi KJ, Haider MF, Khan MA. Quality by design newer technique for pharmaceutical product development. Intelligent Pharmacy. 2024;2:122-129. DOI: https://doi.org/10.1016/j.ipha.2023.10.004
Kaplan ABU, Öztürk N, Çetin M, Vural I, Özer TÖ. The nanosuspension formulations of daidzein: Preparation and in vitro characterization. Turk J Pharm Sci. 2022;19:84. doi: 10.4274/tjps.galenos.2021.81905. DOI: https://doi.org/10.4274/tjps.galenos.2021.81905
Patel GV, Patel VB, Pathak A, Rajput SJ. Nanosuspension of efavirenz for improved oral bioavailability. Drug Dev Ind Pharm. 2014;40:80-91. doi: 10.3109/03639045.2012.746362. DOI: https://doi.org/10.3109/03639045.2012.746362
Pandita D, Ahuja A, Velpandian T, Lather V, Dutta T, Khar R. Characterization and in vitro assessment of paclitaxel loaded lipid nanoparticles. Pharmazie. 2009;64:301-310. PMID: 19530440.
Almalik A, Alradwan I, Kalam MA, Alshamsan A. Effect of cryoprotection on particle size stability of chitosan nanoparticles. Saudi Pharm J. 2017;25:861-867. doi: 10.1016/j.jsps.2016.12.008. DOI: https://doi.org/10.1016/j.jsps.2016.12.008
Townley ER. Griseofulvin. In: Florey K, editor. Analytical Profiles of Drug Substances. Vol.8. Academic Press; 1979. p. 219-249. DOI: https://doi.org/10.1016/S0099-5428(08)60119-7
Zahraa AA, Fatima JJ. Preparation and evaluation of Lercanidipine HCl nanosuspension to improve dissolution rate. Iraqi J Pharm Sci. 2025;33:20-30. doi: 10.31351/vol33iss4SIpp20-30. DOI: https://doi.org/10.31351/vol33iss(4SI)pp20-30
Kral O, Ilbasmis-Tamer S, Han S, Tirnaksiz F. Development of dermal lidocaine nanosuspension formulation by wet milling method. ACS Omega. 2024;9:52:50992-51008. DOI: https://doi.org/10.1021/acsomega.4c05296
Alshweiat A, Katona G, Csóka I, Ambrus R. Design and characterization of loratadine nanosuspension prepared by ultrasonic-assisted precipitation. Eur J Pharm Sci. 2018;122:94-104. doi: 10.1016/j.ejps.2018.06.010. DOI: https://doi.org/10.1016/j.ejps.2018.06.010
Rashed M, Dadashzadeh S, Bolourchian N. The impact of process and formulation parameters on fabrication of Efavirenz nanosuspension. Iran J Pharm Res. 2022;21:e129409. doi: 10.5812/ijpr-129409. DOI: https://doi.org/10.5812/ijpr-129409
Ambrus R, Alshweiat A, Szabó-Révész P, Bartos C, Csóka I. Smartcrystals for efficient dissolution of poorly water-soluble meloxicam. Pharmaceutics. 2022;14:245. doi: 10.3390/pharmaceutics14020245. DOI: https://doi.org/10.3390/pharmaceutics14020245
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Al-Rafidain Journal of Medical Sciences ( ISSN 2789-3219 )

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Published by Al-Rafidain University College. This is an open access journal issued under the CC BY-NC-SA 4.0 license (https://creativecommons.org/licenses/by-nc-sa/4.0/).











