MicroRNAs as a Diagnostic Tool in Bacterial Infections: A Review Article
DOI:
https://doi.org/10.54133/ajms.v10i1.2642Keywords:
Bacterial infection, Extracellular vesicles , microRNA , Sepsis, TuberculosisAbstract
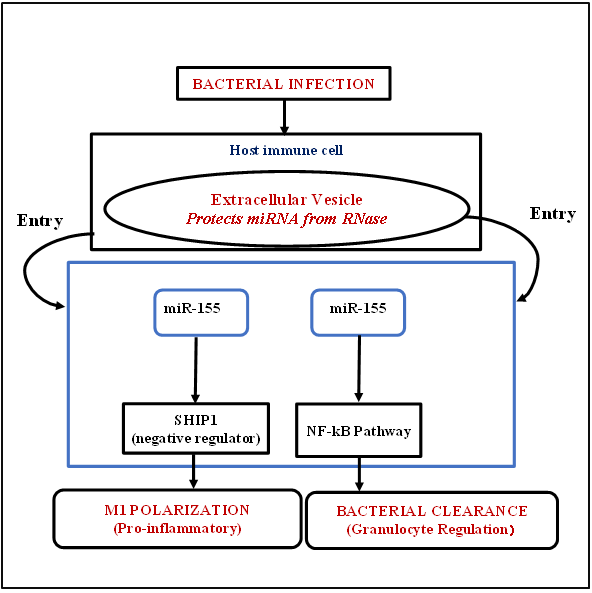
Background: Severe sepsis and tuberculosis are major causes of mortality due to infection all over the world. The impact of microRNAs (miRNAs) in activating the immune process during bacterial infections is essential and associated with managing gene expression of inflammatory markers. Objective: To find the available evidence regarding the diagnostic potential and mechanistic utility of total circulating miRNAs and EV-derived miRNAs in human bacterial infections, that is, sepsis and TB. Methods: This study is based on a PRISMA-DTA literature review. In this study, a comprehensive search was conducted on different databases such as PubMed, Embase, Web of Science, and Scopus between 1 January 2013 and 15 February 2025 for papers related to this topic. The risk of bias was assessed by the QUADAS-2 tool. Results: We evaluated 46 records on sepsis, tuberculosis, severe pneumonia, and bacterial meningitis out of 1,319 records. Most of the studies, about 74%, were case-control using 65% serum, 38% plasma, and 82% qRT-PCR. Sepsis circulating microRNAs showed moderate-to-high diagnostic accuracy, where miR-155-5p AUC=0.81, miR-223 AUC=0.79, and multi-miRNA panels up to AUC=0.90; severe pneumonia showed miR-155 AUC=0.79, miR-34a AUC=0.81, and miR-150 AUC=0.83; tuberculosis showed miR-29a AUC=0.81 and panels including miR-144 and miR-197 AUC=0.87; and bacterial meningitis showed CSF miR-181a AUC=0.82, plasma miR-223 AUC=0.79, and CSF miR-125b AUC=0.84. Conclusions: MicroRNAs in the circulation have demonstrated moderate-to-high forms of diagnostic accuracy among various bacteria and their infections, such as sepsis, tuberculosis, severe pneumonia, and bacterial meningitis.
Downloads
References
Fongang H, Mbaveng AT, Kuete V. Global burden of bacterial infections and drug resistance. Adv Botanical Res. 2023;106:1-20. doi: 10.1016/bs.abr.2022.08.001. DOI: https://doi.org/10.1016/bs.abr.2022.08.001
High KP. Infection as a cause of age-related morbidity and mortality. Ageing Res Rev. 2004;3(1):1-14. doi: 10.1016/j.arr.2003.08.001. DOI: https://doi.org/10.1016/j.arr.2003.08.001
Murray CJ. The global burden of disease study at 30 years. Nat Med. 2022;28(10):2019-2026. doi: 10.1038/s41591-022-01990-1. DOI: https://doi.org/10.1038/s41591-022-01990-1
Ranjit S, Kissoon N. Challenges and solutions in translating sepsis guidelines into practice in resource-limited settings. Transl Pediatr. 2021;10(10):2646. doi: 10.21037/tp-20-310. DOI: https://doi.org/10.21037/tp-20-310
La Via L, Sangiorgio G, Stefani S, Marino A, Nunnari G, Cocuzza S, et al. The global burden of sepsis and septic shock. Epidemiologia. 2024;5(3):456-478. doi: 10.3390/epidemiologia5030032. DOI: https://doi.org/10.3390/epidemiologia5030032
Naghavi M, Vollset SE, Ikuta KS, Swetschinski LR, Gray AP, Wool EE, et al. Global burden of bacterial antimicrobial resistance 1990–2021: a systematic analysis with forecasts to 2050. Lancet. 2024;404(10459):1199-1226. doi: 10.1016/S0140-6736(24)01867-1. DOI: https://doi.org/10.1016/S0140-6736(24)01867-1
Elbehiry A, Marzouk E, Aldubaib M, Abalkhail A, Anagreyyah S, Anajirih N, et al. Helicobacter pylori infection: current status and future prospects on diagnostic, therapeutic and control challenges. Antibiotics. 2023;12(2):191. doi: 10.3390/antibiotics12020191. DOI: https://doi.org/10.3390/antibiotics12020191
Brusselaers N, Kamal HK, Graham D, Engstrand L. Proton pump inhibitors and the risk of gastric cancer: a systematic review, evidence synthesis and life course epidemiology perspective. BMJ Open Gastroenterol. 2025;12(1):e001719. doi: 10.1136/bmjgast-2024-001719. DOI: https://doi.org/10.1136/bmjgast-2024-001719
Bhattacharya S. Early diagnosis of resistant pathogens. Virulence. 2013;4(2):172-184. doi: 10.4161/viru.23326. DOI: https://doi.org/10.4161/viru.23326
Libânio D, Dinis-Ribeiro M, Pimentel-Nunes P. Helicobacter pylori and microRNAs: Relation with innate immunity and progression of preneoplastic conditions. World J Clin Oncol. 2015;6(5):111. doi: 10.5306/wjco.v6.i5.111. DOI: https://doi.org/10.5306/wjco.v6.i5.111
Brook A. Local microRNAs in peritoneal dialysis-related peritonitis: Cardiff University; 2019.
Burgelman M, Vandendriessche C, Vandenbroucke RE. Extracellular vesicles: A double-edged sword in sepsis. Pharmaceuticals. 2021;14(8):829. doi: 10.3390/ph14080829. DOI: https://doi.org/10.3390/ph14080829
Raeven P, Zipperle J, Drechsler S. Extracellular vesicles as markers and mediators in sepsis. Theranostics. 2018;8(12):3348. doi: 10.7150/thno.23453. DOI: https://doi.org/10.7150/thno.23453
Quaglia M, Fanelli V, Merlotti G, Costamagna A, Deregibus MC, Marengo M, et al. Dual role of extracellular vesicles in sepsis-associated kidney and lung injury. Biomedicines. 2022;10(10):2448. doi: 10.3390/biomedicines10102448. DOI: https://doi.org/10.3390/biomedicines10102448
Li C, Sun X, Yang X, Zhang R, Chen J, Wang X. miRNA sequencing identifies immune-associated miRNAs and highlights the role of miR-193b-5p in sepsis and septic shock progression. Sci Rep. 2025;15(1):5323. doi: 10.1038/s41598-025-89946-6. DOI: https://doi.org/10.1038/s41598-025-89946-6
Liu X, Wang W, Bai Y, Zhang H, Zhang S, He L, et al. Identification of a genome-wide serum microRNA expression profile as potential noninvasive biomarkers for chronic kidney disease using next-generation sequencing. J Int Med Res. 2020;48(12):0300060520969481. doi: 10.1177/0300060520969481. DOI: https://doi.org/10.1177/0300060520969481
Lui A, Do T, Alzayat O, Yu N, Phyu S, Santuya HJ, et al. Tumor suppressor MicroRNAs in clinical and preclinical trials for neurological disorders. Pharmaceuticals. 2024;17(4):426. doi: 10.3390/ph17040426. DOI: https://doi.org/10.3390/ph17040426
Mourenza Á, Lorente-Torres B, Durante E, Llano-Verdeja J, Aparicio JF, Fernández-López A, et al. Understanding microRNAs in the context of infection to find new treatments against human bacterial pathogens. Antibiotics. 2022;11(3):356. doi: 10.3390/antibiotics11030356. DOI: https://doi.org/10.3390/antibiotics11030356
Holla S, Balaji KN. Epigenetics and miRNA during bacteria-induced host immune responses. Epigenomics. 2015;7(7):1197-212. doi: 10.2217/epi.15.75. DOI: https://doi.org/10.2217/epi.15.75
Drury RE, O’Connor D, Pollard AJ. The clinical application of microRNAs in infectious disease. Front Immunol. 2017;8:1182. doi: 10.3389/fimmu.2017.01182. DOI: https://doi.org/10.3389/fimmu.2017.01182
Bindayna K. MicroRNA as sepsis biomarkers: a comprehensive review. Int J Mol Sci. 2024;25(12):6476. doi: 10.3390/ijms25126476. DOI: https://doi.org/10.3390/ijms25126476
Correia CN, Nalpas NC, McLoughlin KE, Browne JA, Gordon SV, MacHugh DE, et al. Circulating microRNAs as potential biomarkers of infectious disease. Front Immunol. 2017;8:118. doi: 10.3389/fimmu.2017.00118. DOI: https://doi.org/10.3389/fimmu.2017.00118
Tribolet L, Kerr E, Cowled C, Bean AG, Stewart CR, Dearnley M, et al. MicroRNA biomarkers for infectious diseases: from basic research to biosensing. Front Microbiol. 2020;11:1197. doi: 10.3389/fmicb.2020.01197. DOI: https://doi.org/10.3389/fmicb.2020.01197
Ho PT, Clark IM, Le LT. MicroRNA-based diagnosis and therapy. Int J Mol Sci. 2022;23(13):7167. doi: 10.3390/ijms23137167. DOI: https://doi.org/10.3390/ijms23137167
Rupaimoole R, Slack FJ. MicroRNA therapeutics: towards a new era for the management of cancer and other diseases. Nat Rev Drug Discov. 2017;16(3):203-222. doi: 10.1038/nrd.2016.246. DOI: https://doi.org/10.1038/nrd.2016.246
Dong H, Lei J, Ding L, Wen Y, Ju H, Zhang X. MicroRNA: function, detection, and bioanalysis. Chem Rev. 2013;113(8):6207-6233. doi: 10.1021/cr300362f. DOI: https://doi.org/10.1021/cr300362f
Hu J, Li W, Xie S, Liao Y, Chen T, Wang X, et al. Unveiling neurogenic biomarkers for the differentiation between sepsis patients with or without encephalopathy: an updated meta-analysis. Syst Rev. 2025;14(1):38. doi: 10.1186/s13643-025-02784-5. DOI: https://doi.org/10.1186/s13643-025-02784-5
Zhou X, Li X, Wu M. miRNAs reshape immunity and inflammatory responses in bacterial infection. Signal Transduct Target Ther. 2018;3(1):14. doi: 10.1038/s41392-018-0006-9. DOI: https://doi.org/10.1038/s41392-018-0006-9
Gunasekaran H, Sampath P, Thiruvengadam K, Malaisamy M, Ramasamy R, Ranganathan UD, et al. A systematic review and meta-analysis of circulating serum and plasma microRNAs in TB diagnosis. BMC Infect Dis. 2024;24(1):402. doi: 10.1186/s12879-024-09232-0. DOI: https://doi.org/10.1186/s12879-024-09232-0
Daniel EA, Thiruvengadam K, Chandrasekaran P, Hilda N, Umashankar P, Prashanthi P, et al. Discovery of a blood-based miRNA signature that can predict onset of active tuberculosis among household contacts of TB patients. Front Tuberculosis. 2024;2:1415346. doi: 10.3389/ftubr.2024.1415346. DOI: https://doi.org/10.3389/ftubr.2024.1415346
Kimura M, Kothari S, Gohir W, Camargo JF, Husain S. MicroRNAs in infectious diseases: potential diagnostic biomarkers and therapeutic targets. Clin Microbiol Rev. 2023;36(4):e00015-23. doi: 10.1128/cmr.00015-23. DOI: https://doi.org/10.1128/cmr.00015-23
Wiernsperger N, Al-Salameh A, Cariou B, Lalau J-D. Protection by metformin against severe Covid-19: An in-depth mechanistic analysis. Diabetes Metab. 2022;48(4):101359. doi: 10.1016/j.diabet.2022.101359. DOI: https://doi.org/10.1016/j.diabet.2022.101359
Antonakos N, Gilbert C, Theroude C, Schrijver IT, Roger T. Modes of action and diagnostic value of miRNAs in sepsis. Front Immunol. 2022;13:951798. doi: 10.3389/fimmu.2022.951798. DOI: https://doi.org/10.3389/fimmu.2022.951798
Gaál Z. Role of microRNAs in immune regulation with translational and clinical applications. Int J Mol Sci. 2024;25(3):1942. doi: 10.3390/ijms25031942. DOI: https://doi.org/10.3390/ijms25031942
Abd-El-Fattah AA, Sadik NAH, Shaker OG, Aboulftouh ML. Differential microRNAs expression in serum of patients with lung cancer, pulmonary tuberculosis, and pneumonia. Cell Biochem Biophys. 2013;67(3):875-884. doi: 10.1007/s12013-013-9575-y. DOI: https://doi.org/10.1007/s12013-013-9575-y
Ashirbekov Y, Khamitova N, Satken K, Abaildayev A, Pinskiy I, Yeleussizov A, et al. Circulating microRNAs as biomarkers for the early diagnosis of lung cancer and its differentiation from tuberculosis. Diagnostics. 2024;14(23):2684. doi: 10.3390/diagnostics14232684. DOI: https://doi.org/10.3390/diagnostics14232684
Milhelm Z, Chiroi P, Harangus A, Dudea M, Ciocan C, Pop L, et al. Understanding microRNAs in the context of bacterial versus viral infections. Med Pharm Rep. 2024;97(4):438. doi: 10.15386/mpr-2817. DOI: https://doi.org/10.15386/mpr-2817
Aguilar C, Mano M, Eulalio A. Multifaceted roles of microRNAs in host-bacterial pathogen interaction. Microbiol Spectr. 2019;7(3):10.1128/microbiolspec.bai-0002-2019. doi: 10.1128/9781555819323.ch17. DOI: https://doi.org/10.1128/microbiolspec.BAI-0002-2019
Alı PSS. Role of miRNAs in immune regulation and bacterial infections. J Microbiol Infect Dis. 2023;13(01):1-7. doi: 10.5799/jmid.1264855. DOI: https://doi.org/10.5799/jmid.1264855
Srinivasan R, Ramadoss R, Kandasamy V, Ranganadin P, Green SR, Kasirajan A, et al. Exploring the regulatory role of small RNAs in modulating host-pathogen interactions: Implications for bacterial and viral infections. Mol Biol Rep. 2025;52(1):115. doi: 10.1007/s11033-024-10214-3. DOI: https://doi.org/10.1007/s11033-024-10214-3
Bauer AN, Majumdar N, Williams F, Rajput S, Pokhrel LR, Cook PP, et al. MicroRNAs: small but key players in viral infections and immune responses to viral pathogens. Biology. 2023;12(10):1334. doi: 10.3390/biology12101334. DOI: https://doi.org/10.3390/biology12101334
O'connell RM, Rao DS, Chaudhuri AA, Baltimore D. Physiological and pathological roles for microRNAs in the immune system. Nat Rev Immunol. 2010;10(2):111-122. doi: 10.1038/nri2708. DOI: https://doi.org/10.1038/nri2708
Chandan K, Gupta M, Sarwat M. Role of host and pathogen-derived microRNAs in immune regulation during infectious and inflammatory diseases. Front Immunol. 2020;10:3081. doi: 10.3389/fimmu.2019.03081. DOI: https://doi.org/10.3389/fimmu.2019.03081
Wu Xq, Dai Y, Yang Y, Huang C, Meng Xm, Wu Bm, et al. Emerging role of micro RNA s in regulating macrophage activation and polarization in immune response and inflammation. Immunology. 2016;148(3):237-248. doi: 10.1111/imm.12608. DOI: https://doi.org/10.1111/imm.12608
Mussack V. The role of microRNAs and extracellular vesicles in the detection of autologous blood doping: Technische Universität München; 2022. Available at: https://mediatum.ub.tum.de/doc/1615908/document.pdf
Formosa A, Turgeon P, Dos Santos CC. Role of miRNA dysregulation in sepsis. Mol Med. 2022;28(1):99. doi: 10.1186/s10020-022-00527-z. DOI: https://doi.org/10.1186/s10020-022-00527-z
Yong YK, Tan HY, Saeidi A, Wong WF, Vignesh R, Velu V, et al. Immune biomarkers for diagnosis and treatment monitoring of tuberculosis: current developments and future prospects. Front Microbiol. 2019;10:2789. doi: 10.3389/fmicb.2019.02789. DOI: https://doi.org/10.3389/fmicb.2019.02789
van Ruler D. The role of micro-RNA in the diagnosis of HIV-associated immune reconstitution inflammatory syndrome: University of Pretoria; 2024. doi: 10.25403/UPresearchdata.28435820.
Salameh JP, Bossuyt PM, McGrath TA, Thombs BD, Hyde CJ, Macaskill P, et al. Preferred reporting items for systematic review and meta-analysis of diagnostic test accuracy studies (PRISMA-DTA): explanation, elaboration, and checklist. BMJ. 2020;370. doi: 10.1136/bmj.m2632. DOI: https://doi.org/10.1136/bmj.m2632
Salameh J-P, Moher D, McGrath TA, Frank RA, Sharifabadi AD, Islam N, et al. Assessing adherence to the PRISMA-DTA guideline in diagnostic test accuracy systematic reviews: A five-year follow-up analysis. J Appl Lab Med. 2025;10(2):416-431. doi: 10.1093/jalm/jfae117. DOI: https://doi.org/10.1093/jalm/jfae117
Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, et al. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011;155(8):529-536. doi: 10.7326/0003-4819-155-8-201110180-00009. DOI: https://doi.org/10.7326/0003-4819-155-8-201110180-00009
Lee J, Mulder F, Leeflang M, Wolff R, Whiting P, Bossuyt PM. QUAPAS: an adaptation of the QUADAS-2 tool to assess prognostic accuracy studies. Ann Intern Med. 2022;175(7):1010-1018. doi: 10.7326/M22-0276. DOI: https://doi.org/10.7326/M22-0276
Carra MC, Romandini P, Romandini M. Risk of bias evaluation of cross‐sectional studies: Adaptation of the Newcastle‐Ottawa Scale. J Periodont Res. 2025. doi: 10.1111/jre.13405. DOI: https://doi.org/10.1111/jre.13405
Lo CKL, Mertz D, Loeb M. Newcastle-Ottawa Scale: comparing reviewers’ to authors’ assessments. BMC Med Res Methodol. 2014;14(1):45. doi: 10.1186/1471-2288-14-45. DOI: https://doi.org/10.1186/1471-2288-14-45
Cooper H, Hedges LV, Valentine JC. The handbook of research synthesis and meta-analysis: Russell Sage Foundation; 2019. Available at: https://www.russellsage.org/publications/book/handbook-research-synthesis-and-meta-analysis DOI: https://doi.org/10.7758/9781610448864
Yang B, Mallett S, Takwoingi Y, Davenport CF, Hyde CJ, Whiting PF, et al. QUADAS-C: a tool for assessing risk of bias in comparative diagnostic accuracy studies. Ann Intern Med. 2021;174(11):1592-1599. doi: 10.7326/M21-2234. DOI: https://doi.org/10.7326/M21-2234
Jiang K, Yang J, Guo S, Zhao G, Wu H, Deng G. Peripheral circulating exosome-mediated delivery of miR-155 as a novel mechanism for acute lung inflammation. Mol Ther. 2019;27(10):1758-1771. doi: 10.1016/j.ymthe.2019.07.003. DOI: https://doi.org/10.1016/j.ymthe.2019.07.003
Benz F, Roy S, Trautwein C, Roderburg C, Luedde T. Circulating microRNAs as biomarkers for sepsis. Int J Mol Sci. 2016;17(1):78. doi: 10.3390/ijms17010078. DOI: https://doi.org/10.3390/ijms17010078
Izzotti A, Carozzo S, Pulliero A, Zhabayeva D, Ravetti JL, Bersimbaev R. Extracellular MicroRNA in liquid biopsy: applicability in cancer diagnosis and prevention. Am J Cancer Res. 2016;6(7):1461-93. PMID: 27508091.
He X, Park S, Chen Y, Lee H. Extracellular vesicle-associated miRNAs as a biomarker for lung cancer in liquid biopsy. Front Mol Biosci. 2021;8:630718. doi: 10.3389/fmolb.2021.630718. DOI: https://doi.org/10.3389/fmolb.2021.630718
Xiao Y, Liang J, Witwer KW, Zhang Y, Wang Q, Yin H. Extracellular vesicle-associated microRNA-30b-5p activates macrophages through the SIRT1/NF-κB pathway in cell senescence. Front Immunol. 2022;13:955175. doi: 10.3389/fimmu.2022.955175. DOI: https://doi.org/10.3389/fimmu.2022.955175
do Nascimento MF, Ferreira LRP, Vieira Junior JM, Deheinzelin D, Aparecida Santos Nussbaum AC, Toshihiro Sakamoto LH, et al. Circulating extracellular vesicles as potential biomarkers and mediators of acute respiratory distress syndrome in sepsis. Sci Rep. 2025;15(1):5512. doi: 10.1038/s41598-025-89783-7. DOI: https://doi.org/10.1038/s41598-025-89783-7
Dos Santos CC, Lopes-Pacheco M, English K, Rolandsson Enes S, Krasnodembskaya A, Rocco PR. The MSC-EV-microRNAome: a perspective on therapeutic mechanisms of action in sepsis and ARDS. Cells. 2024;13(2):122. doi: 10.3390/cells13020122. DOI: https://doi.org/10.3390/cells13020122
Giannubilo SR, Cecati M, Marzioni D, Ciavattini A. Circulating miRNAs and preeclampsia: From implantation to epigenetics. Int J Mol Sci. 2024;25(3):1418. doi: 10.3390/ijms25031418. DOI: https://doi.org/10.3390/ijms25031418
Zhao Y, Zhu R, Hu X. Diagnostic capacity of miRNAs in neonatal sepsis: a systematic review and meta-analysis. J Maternal Fetal Neonat Med. 2024;37(1):2345850. doi: 10.1080/14767058.2024.2345850. DOI: https://doi.org/10.1080/14767058.2024.2345850
Li X, Xu Y, Liao P. Diagnostic performance of microRNA-29a in active pulmonary tuberculosis: a systematic review and meta-analysis. Clinics. 2023;78:100290. doi: 10.1016/j.clinsp.2023.100290. DOI: https://doi.org/10.1016/j.clinsp.2023.100290
He J, Xiong J, Huang Y. miR-29 as diagnostic biomarkers for tuberculosis: a systematic review and meta-analysis. Front Public Health. 2024;12:1384510. doi: 10.3389/fpubh.2024.1384510. DOI: https://doi.org/10.3389/fpubh.2024.1384510
Mavridis K, Gueugnon F, Petit-Courty A, Courty Y, Barascu A, Guyetant S, et al. The oncomiR miR-197 is a novel prognostic indicator for non-small cell lung cancer patients. Br J Cancer. 2015;112(9):1527-1535. doi: 10.1038/bjc.2015.119. DOI: https://doi.org/10.1038/bjc.2015.119
Wang C, Ye X, Xu J, Li Q, Sun M, Yi J. Systematic review and meta-analysis of dysregulated miRNAs in patients with severe pneumonia. Turk J Biochem. 2025(0). doi: 10.1515/tjb-2024-0310. DOI: https://doi.org/10.1515/tjb-2024-0310
Wang ZF, Yang YM, Fan H. Diagnostic value of miR-155 for acute lung injury/acute respiratory distress syndrome in patients with sepsis. J Int Med Res. 2020;48(7):0300060520943070. doi: 10.1177/0300060520943070. DOI: https://doi.org/10.1177/0300060520943070
Wang X, Guo P, Tian J, Li J, Yan N, Zhao X, et al. LncRNA GAS5 participates in childhood pneumonia by inhibiting cell apoptosis and promoting SHIP-1 expression via downregulating miR-155. BMC Pulm Med. 2021;21(1):362. doi: 10.1186/s12890-021-01724-y. DOI: https://doi.org/10.1186/s12890-021-01724-y
Zhang C, Sun H, Zhang Q-Y, Tong Z-H. MiR-150 levels are related to in-hospital mortality in non-HIV Pneumocystis pneumonia patients. Med Mycol. 2024;62(5):myae022. doi: 10.1093/mmy/myae022. DOI: https://doi.org/10.1093/mmy/myae022
Huang S, Feng C, Zhai YZ, Zhou X, Li B, Wang LL, et al. Identification of miRNA biomarkers of pneumonia using RNA‑sequencing and bioinformatics analysis. Exp Ther Med. 2017;13(4):1235-1244. doi: 10.3892/etm.2017.4151. DOI: https://doi.org/10.3892/etm.2017.4151
Li M, Lu L, Xu H. Diagnostic value of miR-34a in Mycoplasma pneumoniae pneumonia in children and its correlation with rehabilitation effect. J Cardiothoracic Surg. 2024;19(1):507. doi: 10.1186/s13019-024-02992-5. DOI: https://doi.org/10.1186/s13019-024-02992-5
Ding L, Jiang Y. Biomarkers associated with the diagnosis and prognosis of Mycoplasma pneumoniae pneumonia in children: a review. Front Cell Infect Microbiol. 2025;15:1552144. doi: 10.3389/fcimb.2025.1552144. DOI: https://doi.org/10.3389/fcimb.2025.1552144
Haroun RA-H, Osman WH, Amin RE, Hassan AK, Abo-Shanab WS, Eessa AM. Circulating plasma miR-155 is a potential biomarker for the detection of SARS-CoV-2 infection. Pathology. 2022;54(1):104-110. doi: 10.1016/j.pathol.2021.09.006. DOI: https://doi.org/10.1016/j.pathol.2021.09.006
Gaytán-Pacheco N, Ibáñez-Salazar A, Herrera-Van Oostdam AS, Oropeza-Valdez JJ, Magaña-Aquino M, Adrián López J, et al. miR-146a, miR-221, and miR-155 are involved in inflammatory immune response in severe COVID-19 patients. Diagnostics. 2022;13(1):133. doi: 10.3390/diagnostics13010133. DOI: https://doi.org/10.3390/diagnostics13010133
Backes C, Meese E, Keller A. Specific miRNA disease biomarkers in blood, serum and plasma: challenges and prospects. Mol Diag Ther. 2016;20(6):509-518. doi: 10.1007/s40291-016-0221-4. DOI: https://doi.org/10.1007/s40291-016-0221-4
Szczepanek J. Role of microRNA dysregulation in childhood acute leukemias: Diagnostics, monitoring and therapeutics: A comprehensive review. World J Clin Oncol. 2020;11(6):348. doi: 10.5306/wjco.v11.i6.348. DOI: https://doi.org/10.5306/wjco.v11.i6.348
Rashed WM, Hammad AM, Saad AM, Shohdy KS. MicroRNA as a diagnostic biomarker in childhood acute lymphoblastic leukemia; systematic review, meta-analysis and recommendations. Crit Rev Oncol Hematol. 2019;136:70-78. doi: 10.1016/j.critrevonc.2019.02.008. DOI: https://doi.org/10.1016/j.critrevonc.2019.02.008
Egyed B, Kutszegi N, Sági JC, Gézsi A, Rzepiel A, Visnovitz T, et al. MicroRNA-181a as novel liquid biopsy marker of central nervous system involvement in pediatric acute lymphoblastic leukemia. J Transl Med. 2020;18(1):250. doi: 10.1186/s12967-020-02415-8. DOI: https://doi.org/10.1186/s12967-020-02415-8
Baldassarre A, Felli C, Prantera G, Masotti A. Circulating microRNAs and bioinformatics tools to discover novel diagnostic biomarkers of pediatric diseases. Genes. 2017;8(9):234. doi: 10.3390/genes8090234. DOI: https://doi.org/10.3390/genes8090234
Kelly E, Whelan SO, Harriss E, Murphy S, Pollard AJ, O'Connor D. Systematic review of host genomic biomarkers of invasive bacterial disease: Distinguishing bacterial from non-bacterial causes of acute febrile illness. EBioMedicine. 2022;81. doi: 10.1016/j.ebiom.2022.104110. DOI: https://doi.org/10.1016/j.ebiom.2022.104110
Fejes Z, Erdei J, Pócsi M, Takai J, Jeney V, Nagy A, et al. Elevated pro-inflammatory cell-free microRNA levels in cerebrospinal fluid of premature infants after intraventricular hemorrhage. Int J Mol Sci. 2020;21(18):6870. doi: 10.3390/ijms21186870. DOI: https://doi.org/10.3390/ijms21186870
Péterffy B, Nádasi TJ, Krizsán S, Horváth A, Márk Á, Barna G, et al. Digital PCR-based quantification of miR-181a in the cerebrospinal fluid aids patient stratification in pediatric acute lymphoblastic leukemia. Sci Rep. 2024;14(1):28556. doi: 10.1038/s41598-024-79733-0. DOI: https://doi.org/10.1038/s41598-024-79733-0
Zhong B, Ma S, Wang DH. Ablation of TRPV1 abolishes salicylate-induced sympathetic activity suppression and exacerbates salicylate-induced renal dysfunction in diet-induced obesity. Cells. 2021;10(5):1234. doi: 10.3390/cells10051234. DOI: https://doi.org/10.3390/cells10051234
Peng C, Wang J, Gao W, Huang L, Liu Y, Li X, et al. Meta-analysis of the diagnostic performance of circulating micrornas for pancreatic cancer. Int J Med Sci. 2021;18(3):660. doi: 10.7150/ijms.52706. DOI: https://doi.org/10.7150/ijms.52706
Drokow EK, Sun K, Ahmed HAW, Akpabla GS, Song J, Shi M. Circulating microRNA as diagnostic biomarkers for haematological cancers: a systematic review and meta-analysis. Cancer Manag Res. 2019:4313-4326. doi: 10.2147/CMAR.S199126. DOI: https://doi.org/10.2147/CMAR.S199126
Alemayehu E, Belete MA, Walle M, Getu F, Mulatie Z, Teshome M, et al. Diagnostic accuracy of circulating miRNAs to discriminate hepatocellular carcinoma from liver cirrhosis: a systematic review and meta-analysis. Front Med. 2024;11:1359414. doi: 10.3389/fmed.2024.1359414. DOI: https://doi.org/10.3389/fmed.2024.1359414
Li J, Ma L, Yu H, Yao Y, Xu Z, Lin W, et al. MicroRNAs as potential biomarkers for the diagnosis of chronic kidney disease: a systematic review and meta-analysis. Front Med. 2022;8:782561. doi: 10.3389/fmed.2021.782561. DOI: https://doi.org/10.3389/fmed.2021.782561
Alivernini S, Gremese E, McSharry C, Tolusso B, Ferraccioli G, McInnes IB, et al. MicroRNA-155—at the critical interface of innate and adaptive immunity in arthritis. Front Immunol. 2018;8:1932. doi: 10.3389/fimmu.2017.01932. DOI: https://doi.org/10.3389/fimmu.2017.01932
Nazari-Jahantigh M, Wei Y, Noels H, Akhtar S, Zhou Z, Koenen RR, et al. MicroRNA-155 promotes atherosclerosis by repressing Bcl6 in macrophages. J Clin Investig. 2012;122(11):4190-4202. doi: 10.1172/JCI61716. DOI: https://doi.org/10.1172/JCI61716
Wang Y, Liu X, Xia P, Li Z, FuChen X, Shen Y, et al. The regulatory role of microRNAs on phagocytes: a potential therapeutic target for chronic diseases. Front Immunol. 2022;13:901166. doi: 10.3389/fimmu.2022.901166. DOI: https://doi.org/10.3389/fimmu.2022.901166
Yuan S, Wu Q, Wang Z, Che Y, Zheng S, Chen Y, et al. miR-223: an immune regulator in infectious disorders. Front Immunol. 2021;12:781815. doi: 10.3389/fimmu.2021.781815. DOI: https://doi.org/10.3389/fimmu.2021.781815
Sinigaglia A, Peta E, Riccetti S, Venkateswaran S, Manganelli R, Barzon L. Tuberculosis-associated microRNAs: from pathogenesis to disease biomarkers. Cells. 2020;9(10):2160. doi: 10.3390/cells9102160. DOI: https://doi.org/10.3390/cells9102160
Afum-Adjei Awuah A, Ueberberg B, Owusu-Dabo E, Frempong M, Jacobsen M. Dynamics of T-cell IFN-γ and miR-29a expression during active pulmonary tuberculosis. Int Immunol. 2014;26(10):579-582. doi: 10.1093/intimm/dxu068. DOI: https://doi.org/10.1093/intimm/dxu068
Kim JK, Lee HM, Park KS, Shin DM, Kim TS, Kim YS, et al. MIR144* inhibits antimicrobial responses against Mycobacterium tuberculosis in human monocytes and macrophages by targeting the autophagy protein DRAM2. Autophagy. 2017;13(2):423-441. doi: 10.1080/15548627.2016.1241922. DOI: https://doi.org/10.1080/15548627.2016.1241922
Liu Y, Wang R, Jiang J, Yang B, Cao Z, Cheng X. miR-223 is upregulated in monocytes from patients with tuberculosis and regulates function of monocyte-derived macrophages. Mol Immunol. 2015;67(2):475-481. doi: 10.1016/j.molimm.2015.08.006. DOI: https://doi.org/10.1016/j.molimm.2015.08.006
Behrouzi A, Alimohammadi M, Nafari AH, Yousefi MH, Riazi Rad F, Vaziri F, et al. The role of host miRNAs on Mycobacterium tuberculosis. ExRNA. 2019;1(1):40. doi: 10.1186/s41544-019-0040-y. DOI: https://doi.org/10.1186/s41544-019-0040-y
Harris T, Davenport R, Hurst T, Jones J. Improving outcome in severe trauma: trauma systems and initial management—intubation, ventilation and resuscitation. Postgrad Medical J. 2012;88(1044):588-594. doi: 10.1136/postgradmedj-2010-74245. DOI: https://doi.org/10.1136/postgradmedj-2010-74245
Dewanto I, Koontongkaew S, Widyanti N. Characteristics of dental services in rural, suburban, and urban areas upon the implementation of indonesia national health insurance. Front Public Health. 2020;8:138. doi: 10.3389/fpubh.2020.00138. DOI: https://doi.org/10.3389/fpubh.2020.00138
Wang H, Zhang P, Chen W, Feng D, Jia Y, Xie L. Serum microRNA-21 is a potential biomarker for sepsis. Int J Clin Exp Med. 2014;7(11):4345-4351. doi: 10.3892/ol.2018.8972. DOI: https://doi.org/10.3892/ol.2018.8972
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Al-Rafidain Journal of Medical Sciences ( ISSN 2789-3219 )

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Published by Al-Rafidain University College. This is an open access journal issued under the CC BY-NC-SA 4.0 license (https://creativecommons.org/licenses/by-nc-sa/4.0/).











